Amylase is an example of:
1.
Oxidoreductase
2.
Transferase
3.
Hydrolase
4.
Ligase
The figure given below shows the conversion of a substrate into product by an enzyme. In which one of the four options (a-d) the components of reaction labelled as A, B, C and D are identified correctly?
|
A |
B |
C |
D |
|
|
(1) |
Potential energy |
Transition state |
Activation energy with enzyme |
Activation energy without enzyme |
|
(2) |
Transition state |
Potential energy |
Activation energy without enzyme |
Activation energy with enzyme |
|
(3) |
Potential energy |
Transition state |
Activation energy with enzyme |
Activation energy without enzyme |
|
(4) |
Activation energy with enzyme |
Transition state |
Activation energy without enzyme |
Potential energy |
The adjoining graph shows change in conc. of substrate on enzyme activity. Identify A, B and C:
|
A |
B |
C |
|
|
1. |
Ki |
Km |
Vmax |
|
2. |
Km |
Ki |
|
|
3. |
Vmax |
Km |
|
|
4. |
Km |
Vmax |
Go through the figures and select the correct option:
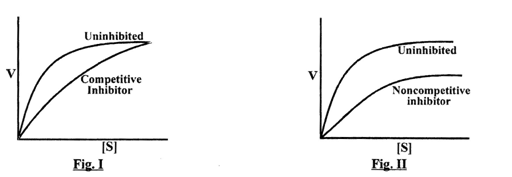
| 1. |
I - In non-competitive type of enzymatic inhibition the V max decreases and Km remain unchanged II - In competitive type of enzymatic inhibition the Vmax decreases and Km remain unchanged |
| 2. |
II - In non-competitive type of enzymatic inhibition the Vmax decreases and Km remain unchanged I - In competitive type of enzymatic inhibition the Vmax remain same and Km is changed |
| 3. |
I - In non-competitive type of enzymatic inhibition the Vmax increases and Km remain unchanged II - In competitive type of enzymatic inhibition the Vmax changes and Km decreases |
| 4. |
I - In non-competitive type of enzymatic inhibition the Vmax increases and Km remain unchanged II - In competitive type of enzymatic inhibition the Vmax increases and Km increases |
Assertion : The amino acid glycine comes under the category of nonessential amino acids.
Reason : This is due to the fact that it can not be synthesised in the body.
- If both the assertion and the reason are true and the reason is a correct explanation of the assertion
- If both the assertion and reason are true but the reason is not a correct explanation of the assertion
- If the assertion is true but the reason is false
- If both the assertion and reason are false
In competitive inhibition:
| 1. | Inhibitor resembles the substrate in molecular structure |
| 2. | Competition between substrates and inhibitors to occupy active sites |
| 3. | Binding the inhibitors to active sites declines the enzyme action |
| 4. | All are correct |
Choose the correct statement(s):
| 1. | Km (Michaelis - Menten) constant is the substrate concentration at which the enzymatic reaction attains half of its maximum velocity (1/2 Vmax) |
| 2. | At lower Km, higher the substrate affinity for enzyme |
| 3. | Vmax is reached when all the active sites of an enzyme are saturated with substrate |
| 4. | All are correct |
Which one of the following graphs show the relationship between the rate of an enzymatic activity and substrate conc.(S):
Which one of the graphs show the effect of temperature on the velocity of a typical enzymatic reaction?
Which one of the graphs shows the effect of pH on the velocity of a typical enzymatic reaction (V)?
1.









