KI in acetone, undergoes SN2 reaction with each of P, Q, R and S, the rates of the reaction vary as:
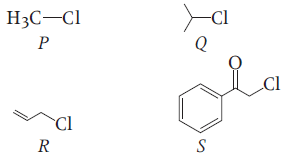
1. P > Q > R > S
2. S > P > R > Q
3. P > R > Q > S
4. R > P > S > Q
In an SN1 reaction on chiral centres there is
1. 100% racemisation
2. inversion more than retention leading to partial racemisation
3. 100% retention
4. 100% inversion
Consider the following bromides:
the correct order of SN1 reactivity is:
1. A>B>C
2. B>C>A
3. B>A>C
4. C>B>A
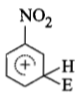
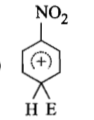
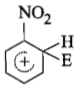
The electrophile. E attacks the benzene ring to generate the intermediate - complex. Of the following which - complex is of lowest energy?
1. 
2. 
3. 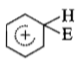
4. 
If X is halogen the correct order for SN2 reactivity is-
1. R2CHX > R3CX > RCH2X
2. RCH2X > R3CX > R2CHX
3. RCH2X > R2CHX > R3X
4. R3CX > R2CHX > RCH2X
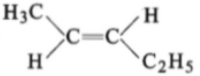
The compound which reacts with HBr obeying Markownikoff's rule is:
1.
2. 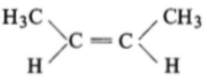
3. 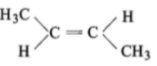
4. 
The formation of cyanohydrin from a ketone is an example of:
(1) electrophilic addition
(2) nucleophilic addition
(3) nucleophilic substitution
(4) electrophilic substitution
Which of the following is an example of the elimination reaction?
1. Chlorination of methane
2. Dehydration of ethanol
3. Nitration of benzene
4. Hydroxylation of ethylene
The SN2 mechanism for R-X + KOH (aq) R-OH + KX follows with:
(1) 100% inversion
(2) 50% inversion
(3) 40% inversion
(4) 30% inversion
The elimination reaction among the following is:
1. CH3CH3+Cl2CH3CH2Cl+ HCl
2. CH3Cl+ KOH(aq.) CH3OH +KCl
3. CH2=CH2 +Br2 CH2BrCH2Br
4. C2H5Br+ KOH(alc.) C2H4 + KBr+H2O







