The excluded volume of a gas will be larger, if is:
1. Small
2. Large
3. Equal to 1
4. Less than unity
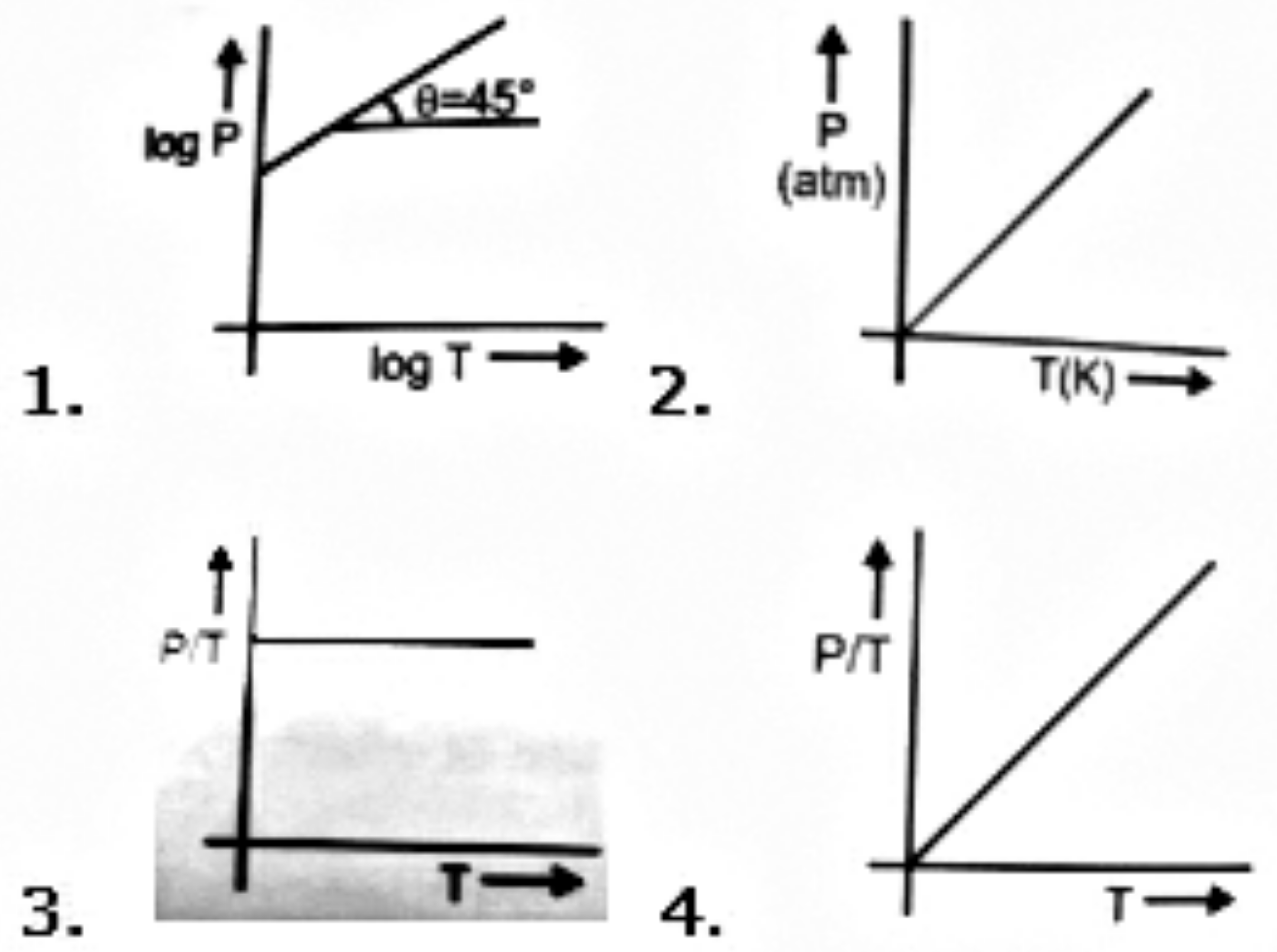
However great the pressure, a gas cannot be liquified above its:
1. Boyle temperature
2. Inversion temperature
3. Critical temperature
4. Room temperature
The van der Waal's equation of law of corresponding states for 1 mole of gas is:
1.
2.
3.
4.
The van der Waals parameters for gases W, X, Y and Z are
|
Gas |
a (atm L2 mol-2) |
b(L mol-1) |
|
W |
4.0 |
0.027 |
|
X |
8.0 |
0.030 |
|
Y |
6.0 |
0.032 |
|
Z |
12.0 |
0.027 |
Which one of these gases has the highest critical temperature?
1. W
2. X
3. Y
4. Z
In two vessels of 1 litre each at the same temperature 1 g of and 1 g of are taken, for these:
1. values will be same
2. Kinetic energy per mol will be same
3. Total kinetic energy will be same
4. Pressure will be same
Two flasks of equal volume connected by a narrow tube (of negligible volume) at 27ºC and contain 0.70 mole of at 0.5 atm. One of the flasks is then immersed into a hot bath, kept at 127ºC, while the other remains at 27ºC. Calculate the final pressure.
(1) 5.714 atm
(2) 0.5714 atm
(3) 2.5214 atm
(4) 5.5114 atm
A gas mixture consists of 2 moles of oxygen and 4 moles of a argon at temperature T. Neglecting all vibrational modes, the total internal energy of the system is –
(A) 4RT
(B) 5RT
(C) 15RT
(D) 11RT
The valves X and Y are opened simultaneously. The white fumes of will first form at :
(A) A
(B) B
(C) C
(D) A, B and C simultaneously
X ml of gas effuses through a hole in a container in 5 seconds. The time taken for the effusion of the same volume of the gas specified below under ideal condition is
(A) 10 seconds :
(B) 20 seconds :
(C) 25 seconds :
(D) 55 seconds :