The figure shows the (P-V) diagram of an ideal gas undergoing a change of state from A to B. Four different paths I, II, III and IV, as shown in the figure, may lead to the same change of state.

(a)
The change in internal energy is the same in cases IV and III but not in cases I and II.
(b)
The change in internal energy is the same in all four cases.
(c)
Work done is maximum in case I.
(d)
Work done is minimum in case II.
Which of the following options contains only correct statements?
1.
(b), (c), (d)
2.
(a), (d)
3.
(b), (c)
4.
(a), (c), (d)

Which of the following options contains only correct statements?
Consider a cycle followed by an engine (figure).
1 to 2 is isothermal,
2 to 3 is adiabatic,
3 to 1 is adiabatic.
Such a process does not exist, because:
| (a) | heat is completely converted to mechanical energy in such a process, which is not possible. |
| (b) | In this process, mechanical energy is completely converted to heat, which is not possible. |
| (c) | curves representing two adiabatic processes don’t intersect. |
| (d) | curves representing an adiabatic process and an isothermal process don't intersect. |
Choose the correct alternatives:
1. (a, b)
2. (a, c)
3. (b, c)
4. (c, d)
The pressure and volume of a gas are changed as shown in the P-V diagram. The temperature of the gas will:
| 1. | increase as it goes from A to B. |
| 2. | increase as it goes from B to C. |
| 3. | remain constant during these changes. |
| 4. | decrease as it goes from D to A. |
Two cylinders contain the same amount of an ideal monoatomic gas. The same amount of heat is given to two cylinders. If the temperature rise in cylinder A is T0, then the temperature rise in cylinder B will be:
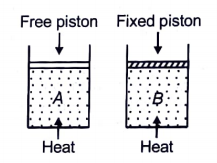
1.
2.
3.
4.
The incorrect relation is:
(where symbols have their usual meanings)
1.
2.
3.
4.
We consider a thermodynamic system. If ∆U represents the increase in its internal energy and W the work done by the system, which of the following statements is true?
| 1. | \(\Delta \mathrm{U}=-\mathrm{W}\) in an isothermal process |
| 2. | \(\Delta \mathrm{U}=\mathrm{W}\) in an isothermal process |
| 3. | \(\Delta \mathrm{U}=-\mathrm{W}\) in an adiabatic process |
| 4. | \(\Delta \mathrm{U}=\mathrm{W}\) in an adiabatic process |
Two cylinders, A and B, of equal capacity are connected to each other via a stopcock. A contains gas at a standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. If the stopcock is suddenly opened, then the change in internal energy of the gas is:
| 1. | 0
|
2. | 5 J
|
| 3. | 1 J
|
4. | 3 J |
Find out the total heat given to diatomic gas in the process ABC : (BC is isothermal)
1.
2.
3.
4. 3
0.04 mole of an ideal monatomic gas is allowed to expand adiabatically so that its temperature changes from 800 K to 500 K. The work done during expansion is nearly equal to:
| 1. | \(129.6\) J | 2. | \(-129.6\) J |
| 3. | \(149.6\) J | 4. | \(-149.6\) J |
If an ideal gas undergoes two processes at constant volumes as shown in the pressure-temperature (P-T) diagram, then:
1. =
2. >
3. <
4.







