The hydrogenation of benzoyl chloride in the presence of Pd and BaSO4 gives:
1. Benzyl alcohol
2. Benzaldehyde
3. Benzoic acid
4. Phenol
An alkene “A” on reaction with O3 and Zn - H2O gives propanone and ethanal in an equimolar ratio. The addition of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is:
| 1. | 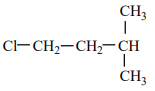 |
2. | 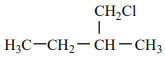 |
| 3. | 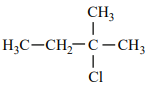 |
4. | 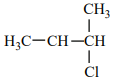 |
What is product 'B' in the below mentioned reaction?
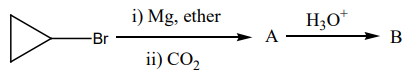
| 1. | 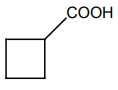 |
2. | 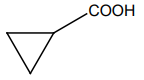 |
| 3. | 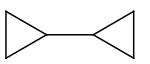 |
4. | 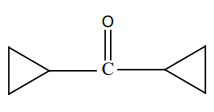 |
The formation of cyanohydrin from acetone is an example of:
1. Nucleophilic substitution.
2. Electrophilic substitution.
3. Electrophilic addition.
4. Nucleophilic addition.
2-Methyl propene on oxidation with hot KMnO4 gives:
1. Acetone
2. Ethanoic acid
3. CO2 and H2O
4. Both 1 & 3
What is the most suitable reagent for the below mentioned conversion?
1. Tollens' reagent
2. Benzoyl peroxide
3. and NaOH solution
4. Sn and NaOH solution
The conversions can be used for Clemmensen reduction are:
a. Benzaldehyde into benzyl alcohol.
b. Cyclohexanone into cyclohexane.
c. Benzoyl chloride into benzaldehyde.
d. Benzophenone into diphenyl methane.
| 1. | a and b | 2. | b and c |
| 3. | c and d | 4. | b and d |
Match the common names given in Column I with the IUPAC names given in Column II.
| Column l (Common names) |
Column ll (IUPAC names) |
| A. Cinnamaldehyde | 1. Pentanal |
| B. Acetophenone | 2. Prop-2-enal |
| C. Valeraldehyde | 3. 1-phenylethanone |
| D. Acrolein | 4. 3-Phenylprop-2-en-al |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 1 | 2 |
Match the acids given in Column I with their correct IUPAC names given in Column II and mark the appropriate option:
| Column l (Acids) |
Column ll (IUPAC names) |
| A. Phthalic acid | 1. Hexane-1,6-dioic acid |
| B. Glutaric acid | 2. Benzene-1,2-dicarboxylic acid |
| C. Succinic acid | 3. Pentane-1,5-dioic acid |
| D. Adipic acid | 4. Butane-1,4-dioic acid |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 2 | 1 |
The reaction that does not give benzoic acid as the major product is:
| 1. |  \(\xrightarrow{K_2Cr_2O_7}\) \(\xrightarrow{K_2Cr_2O_7}\) |
2. |  \(\xrightarrow[(ii)H_3O^+]{(i)NaOCl}\) \(\xrightarrow[(ii)H_3O^+]{(i)NaOCl}\) |
| 3. |  \(\xrightarrow{PCC}\) \(\xrightarrow{PCC}\) |
4. |  \(\xrightarrow{KMnO_4/H^+}\) \(\xrightarrow{KMnO_4/H^+}\) |


