Reason(R): Principal quantum number is a measure of the most possible distance of finding the electron around the nucleus.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
The correct statement(s) among the following regarding quantum numbers is -
| a. | Angular quantum number determines the three-dimensional shape of the orbital |
| b. | The principal quantum number determines the orientation and energy of the orbital |
| c. | Magnetic quantum number determines the size of the orbital |
| d. | Spin quantum number of an electron determines the orientation of the spin of the electron relative to the chosen axis |
1. (a), (d)
2. (b), (c)
3. (c), (d)
4. (b), (d)
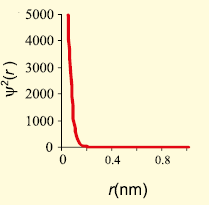
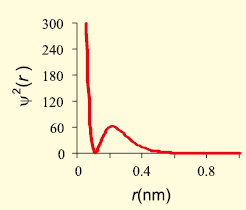
The graph that represents the probability density vs distance of the electron from the nucleus for 2s orbital is -
1.
2.
3.
4.
Match the following:
|
Column I |
Column II |
|
(a) n = 2, l =1 |
i = 4s |
|
(b) n = 4, l = 0 |
ii = 2p |
|
(c) n = 5, l = 3 |
iii = 3d |
|
(d) n = 3, l = 2 |
iv = 5f |
1. a = iv; b = ii; c = i; d = iii
2. a = ii; b = iii; c = iv; d = i
3. a = ii; b = i; c = iv; d = iii
4. a = iii; b = ii; c = i; d = iv
Match the following.
|
Column I |
Column II |
|
A. Photon |
1. Value is 4 for N-shell |
|
B. Electron |
2. Probability density |
|
C. |
3. Always positive value |
|
D. Principle quantum number n |
4. Exhibits both momentum and wavelength |
| Options: | A | B | C | D |
| 1. | 4 | 4 | 2,3 | 1,3 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 2,1 | 4 | 3 | 2,3 |
| 4. | 4 | 5 | 3 | 2 |
Match the quantum numbers with the information provided by them:
| Quantum number | Information provided |
| A. Principal quantum number | 1. Orientation of the orbital |
| B. Azimuthal quantum number | 2. Energy and size of orbital |
| C. Magnetic quantum number | 3. Spin of electron |
| D. Spin quantum number | 4. Shape of the orbital |
Codes
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |