The number of bonds and bonds in the following structure is :
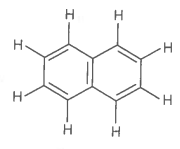
1.
6, 19
2.
4, 20
3.
5, 19
4.
5, 20

The species doesn't have unpaired electrons is:
| 1. | \(N^+_2\) | 2. | \(O_2\) |
| 3. | \(O^{2-}_2\) | 4. | \(B_2\) |
If the electronic configuration of an element is , the four electrons that participate in the chemical bond formation will be from :
1.
2.
3.
4.
The correct bond angle for \(\mathrm{sp}^2\) hybridization is:
| 1. | \(90^\circ\) | 2. | \(120^\circ\) |
| 3. | \(180^\circ\) | 4. | \(109^\circ\) |
The electronic configurations of the elements A, B, and C are given below.
The stable form of A may be represented by the formula:
| 1. | A | 2. | A2 |
| 3. | A3 | 4. | A4 |
The electronic configurations of the elements A, B, and C are given below.
The stable form of C may be represented by the formula :
| 1. | C | 2. | C2 |
| 3. | C3 | 4. | C4 |
The electronic configurations of the elements A, B, and C are given below.
The bond between B and C will be :
| 1. | Ionic. | 2. | Covalent. |
| 3. | Hydrogen. | 4. | Coordinate. |
From the perspective of molecular orbital theory, which statement is false?
1. is not a stable molecule.
2. is not stable but is expected to exist.
3. Bond strength of is maximum amongst the homonuclear diatomic molecules belonging to the second period.
4. The order of energies of molecular orbitals in molecule is:
What is the correct sequence of bond order for the specified species?
| 1. | \(\mathrm{O}^-_2>\mathrm{O}_2>\mathrm{O}^+_2\) | 2. | \(\mathrm{O}^-_2<\mathrm{O}_2<\mathrm{O}^+_2\) |
| 3. | \(\mathrm{O}^-_2>\mathrm{O}_2<\mathrm{O}^+_2\) | 4. | \(\mathrm{O}^-_2<\mathrm{O}_2>\mathrm{O}^+_2\) |
The shape of AsF5 is:
| 1. | Pentagonal bipyramidal. | 2. | Trigonal bipyramidal. |
| 3. | Distorted tetrahedral. | 4. | Square pyramidal. |


