What results from a reaction between concentrated H2SO4 and acetone?
1. Phorone
2. Mesitylene
3. Mesityl oxide
4. Crotonaldehyde
Compounds A and B are, respectively:
| A | B | |
| 1. |  CH2CH2CHO CH2CH2CHO |
 CH=CH-CH2OH CH=CH-CH2OH |
| 2. |  CH2CH2CH2OH CH2CH2CH2OH |
 CH=CH-CH2OH CH=CH-CH2OH |
| 3. |  CH=CH-CH2OH CH=CH-CH2OH |
 CH=CH-CH2OH CH=CH-CH2OH |
| 4. |  CH2CH2CH2OH CH2CH2CH2OH |
 CH2CH2CH2OH CH2CH2CH2OH |


| (i) | (ii) | (iii) | |
| 1. | CH3 – MgI, H3O+ | H2SO4, ∆ | HBr, R2O2 |
| 2. | CH3 – MgI, H3O+ | H2SO4, ∆ | HBr |
| 3. | CH3 – MgI, H3O+ | HBr | |
| 4. | HBr, R2O2 | CH3 – MgI, H3O+ |
The product 'B' in the below mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. | None of these |
CH3–CH2–CHO + dil NaOH gives X
What is product X in the above reaction?
| 1. | |
| 2. | |
| 3. |  |
| 4. |  |
On hydrolysis, a sweet-smelling ester A , with a molar mass of 116, produces a carboxylic acid and an alcohol. Alcohol gives a positive iodoform reaction. The formula for A will be:
| 1. | |
| 2. | CH3CH2–O–COC3H7 |
| 3. | CH3–O–COC4H9 |
| 4. |
The correct statement among the following about HCOOH is:
1. It is a stronger acid than CH3COOH
2. It reduces Tollen’s reagent
3. It gives CO and H2O on heating with conc.H2SO4
4. All of the above
Among the following acids, which one has the smallest dissociation constant?
| 1. |  |
2. |  |
| 3. |  |
4. | 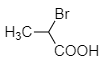 |
A compound that can be reduced to the corresponding hydrocarbon by Zn-Hg/ conc. HCl is:
1. Butan-2-one
2. Acetic acid
3. Acetamide
4. Ethyl acetate
What is the correct order of the boiling point of the below mentioned compound?
| I | CH3CH2CH2CH2OH |
| II | CH3CH2CH2CHO |
| III | CH3CH2CH2COOH |
1. I > II > III
2. III > I > II
3. I > III > II
4. III > II > I




