
The product (B) in the above-mentioned reaction is:
1.

2.

3.

4.






Cyclopentadiene is much more acidic than cyclopentane, beacuse -
1. Cyclopentadiene has conjugated double bonds.
2. Cyclopentadiene has both sp2 and sp3 hybridized carbon atoms.
3. Cyclopentadiene is a strain-free cyclic system.
4. Cyclopentadienyl anion ion, the conjugate base of cyclopentadiene, is an aromatic species and hence has higher stability.
The compounds among the following that can be generated by Friedel craft acylation is/are -
| I. | 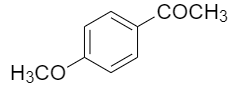 |
| II. |  |
| III. |  |
| IV. |  |
| 1. | II, III and IV | 2. | I, III and IV |
| 3. | I and II | 4. | II and III |
The product B in the above-mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The major product obtained in the given reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The deactivating but ortho-para directing benzene ring substituent among the following is-
1. -N=O
2. -OCH3
3. -COCH3
4. -NO2
The compound X in the above reaction is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The oxidation of benzene by V2O5 in the presence of air produces:
| 1. | Benzoic anhydride | 2. | Maleic anhydride |
| 3. | Benzoic acid | 4. | Benzaldehyde |
A compound among the following that does not undergo Friedel-Craft's reaction easily is-
1. Cumene
2. Xylene
3. Nitrobenzene
4. Toluene
Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO3 and conc.
H2SO4. In the mixture, nitric acid acts as a/an-
1. Reducing agent
2. Acid
3. Base
4. Catalyst





