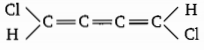Which of the following is not a perfect nonpolar molecule?
1.
2. CCl4
3.
4. All of the above
Choose the correct option for the following molecule:
1. Non planar
2.
3. Both (1) and (2)
4.
The compound that has the highest ionic character associated with the X-Cl bond is:
1.
2.
3.
4.
The least polar bond is present in which of the following?
| 1. | O-F | 2. | P-F |
| 3. | Si-N | 4. | B-F |
The correct order of increasing s-character (in percentage) in the hybrid orbitals of the following molecules/ions is:
| (i) | \(\mathrm{CO}^{2-}_3\) |
| (ii) | \(\mathrm{XeF}_4\) |
| (iii) | \(\mathrm{I}^-_3\) |
| (iv) | \(\mathrm{NCl}_3\) |
| (v) | \(\mathrm{BeCl}_2\) |
| 1. | (ii)<(iii)<(iv)<(i)<(v) | 2. | (ii)<(iv)<(iii)<(v)<(i) |
| 3. | (iii)<(ii)<(i)<(v)<(iv) | 4. | (ii)<(iv)<(iii)<(i)<(v) |
The total number of possible resonance forms for the nitrate ion, () is:
| 1. | 1 | 2. | 2 |
| 3. | 3 | 4. | 4 |
The shape of is:
| 1. | Trigonal planar | 2. | Pyramidal |
| 3. | Bent T-shape | 4. | See-saw |
The molecular geometry that is least likely to result from a trigonal bipyramidal electron geometry is:
| 1. | Trigonal planar | 2. | See-saw |
| 3. | Linear | 4. | T-shaped |
On the left are given decreasing orders of elements for a certain property and on right side is given the property. The incorrect order for the given properties is:
1. : Lattice energy
2. : Electron affinity
3. : Lewis acidic character
4. : Decreasing ionic character
OO bond length ( ) in and is respectively:
| 1. | 1.22 , 1.48 | 2. | 1.48 , 1.22 |
| 3. | 1.22 , 1.22 | 4. | 1.48 , 1.48 |