Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses because:
1.
Alcohols form van der waal bonds with water while hydrocarbons do not
2.
Alcohols form covalent bonds with water while hydrocarbons do not
3.
Alcohols form ionic bonds with water while hydrocarbons do not
4.
Alcohols form H-bonds with water while hydrocarbons do not

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Hydroboration-oxidation reaction is -
1. The addition of borane followed by oxidation
2. The addition of propane followed by oxidation
3. The addition of propane followed by reduction
4. The addition of borane followed by reduction

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The steam-volatile compound x among the following is -
1. o-Nitrophenol
2. p-Nitrophenol
3. Both 1 and 2
4. None of the above

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Ortho nitrophenol is more acidic than ortho methoxy phenol because:
| 1. | The NO2-group increases the electron density in phenol while the methoxy group decreases the electron density in phenol |
| 2. | The nitro-group is an electron-withdrawing group while the methoxy group is an electron-releasing group |
| 3. | The methoxy-group is an electron-withdrawing group while the nitro group is an electron-releasing group |
| 4. | None of the above |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
Preparation of ethers by 2 or 3-degree alcohols in an acidic medium is not a suitable method because:
| 1. | In case of secondary or tertiary alcohols, ketone is obtained as a product. |
| 2. | In case of secondary or tertiary alcohols, aldehyde is obtained as a product. |
| 3. | In case of secondary or tertiary alcohols, alkene is obtained as a product. |
| 4. | In case of primary alcohols, alkene is obtained as a product. |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The IUPAC name of the above compound is:

1. 1-Phenylpropan-2-ol
2. 2-Phenylpropan-2-ol
3. 1-Phenylpropan-1-ol
4. 2-Phenylpropan-1-ol

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The IUPAC name of the below compound is:
1. 3,4-Dimethylhexane –1,3,5-triol
2. 3,5-Dimethylhexane –1,1,5-triol
3. 3,5-Dimethylhexane –1,3,5-triol
4. 3,1-Dimethylhexane –1,3,5-triol

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
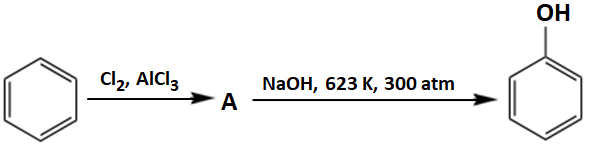
"A" in the reaction below is:
| 1. | Chlorobenzene | 2. | 1,2-Dichlorobenzene |
| 3. | 1,3-dichlorobenzene | 4. | None of these |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
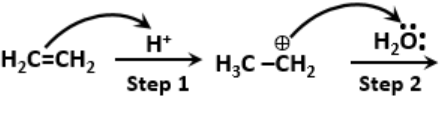
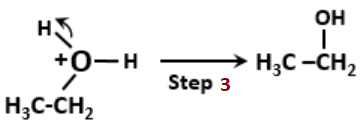
The proposed mechanism for hydration of ethene to yield ethanol is as follows:
The wrong step in the above mechanism is-
1. Step 1
2. Step 2
3. Step 3
4. Both steps 2 and 3

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.
The reaction of 3-methylbutan-2-ol with HBr gives -
| 1. | 2-Bromo-3-methyl butane | 2. | 2-Bromo-2-methyl butane |
| 3. | 3-Bromo-2-methyl butane | 4. | 3-Bromo-3-methyl butane |

To unlock all the explanations of this course, you need to be enrolled.

To unlock all the explanations of this course, you need to be enrolled.