Select Chapter Topics:
How is m-Bromotoluene prepared?
1.
Bromination of toluene
2.
Friedel Craft’s reaction of bromobenzene with CH3Cl
3.
Bromination of nitrobenzene and subsequent replacement of –NO2 group with an ethyl group
4.
Bromination of Aceto–p–toluidine followed by hydrolysis and deamination
Subtopic: Mechanism of Reactions |
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Which of the following molecules can readily undergo the SN2 mechanism?
| 1. | \(\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{Cl} \) | 2. | \(\mathrm{CH}_2=\mathrm{CH}-\mathrm{Cl} \) |
| 3. | \(\mathrm{CH}_2=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CH}_2 \mathrm{Cl}\) | 4. |  |
Subtopic: Mechanism of Reactions |
72%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
Sandmeyer's reaction described by among the following is:
| 1. | |
| 2. | |
| 3. | |
| 4. |  |
Subtopic: Mechanism of Reactions | Chemical Properties |
90%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
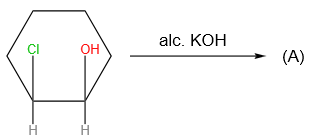
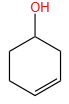
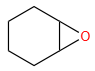
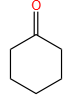
Products (A) and (B) in the given below reactions, respectively, are:

| (A) | (B) | |
| 1. |  |
 |
| 2. |  |
 |
| 3. |  |
 |
| 4. | None of the above | |
Subtopic: Introduction and Physical Properties | Chemical Properties |
57%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
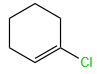
Among the following, which hydrolysis reaction occurs at the slowest rate:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Subtopic: Mechanism of Reactions |
66%
From NCERT
NEET - 2019
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which of the options is correct about product (A)?
1. Product is aromatic
2. Product has a high dipole moment
3. Product has less resonance energy
4. Both A and B
Subtopic: Chemical Properties |
71%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The X and Y in the below mentioned reaction are
| X | Y | |
| 1. | Dilute aqueous NaOH, 20 oC | HBr/Acetic acid, 20 oC |
| 2. | Concentrated alcoholic NaOH, 80 oC | HBr/Acetic acid, 20 oC |
| 3. | Diluted aqueous NaOH, 20 oC, | Br2 / CHCl3 , 0 oC |
| 4. | Concentrated alcoholic NaOH, 80 ° C | Br2/CHCl3, 0 oC |
Subtopic: Chemical Properties |
67%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
What is the total number of chiral products produced by monochlorination of 2-methylbutane ?
1. 2
2. 4
3. 6
4. 8
Subtopic: Chemical Properties | Isomerism & Chirality |
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which of the following reactions cannot form new carbon-carbon bonds?
1. Reimer-Tiemann reaction
2. Cannizaro reaction
3. Wurtz reaction
4. Friedel-Crafts acylation
Subtopic: Mechanism of Reactions |
62%
From NCERT
AIPMT - 2010
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch