The structure of Hex-2-en-4-ynoic acid is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | None of these |
X + CH3COOH → Ester + H2O
| 1. | Alcohol | 2. | Pyridine |
| 3. | Secondary amine | 4. | Acid |
Select the correct option based on the statements below:
| Assertion (A): | The geometry of formaldehyde molecule is planar. |
| Reason (R): | Formaldehyde molecule contains sp2 hybridized carbon atom. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
The reaction that does not give benzoic acid as the major product is:
| 1. |  \(\xrightarrow{K_2Cr_2O_7}\) \(\xrightarrow{K_2Cr_2O_7}\) |
2. |  \(\xrightarrow[(ii)H_3O^+]{(i)NaOCl}\) \(\xrightarrow[(ii)H_3O^+]{(i)NaOCl}\) |
| 3. |  \(\xrightarrow{PCC}\) \(\xrightarrow{PCC}\) |
4. |  \(\xrightarrow{KMnO_4/H^+}\) \(\xrightarrow{KMnO_4/H^+}\) |
The product of the below-mentioned reaction is:
| 1. | 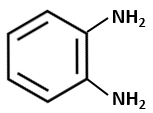 |
2. |  |
| 3. |  |
4. |  |
In the below reaction, product 'P' is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The correct statement among the following about HCOOH is:
1. It is a stronger acid than CH3COOH
2. It reduces Tollen’s reagent
3. It gives CO and H2O on heating with conc.H2SO4
4. All of the above
An acid that forms an anhydride (X) on heating and an acid imide (Y) on strong heating with ammonia is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Match the acids given in Column I with their correct IUPAC names given in Column II and mark the appropriate option:
| Column l (Acids) |
Column ll (IUPAC names) |
| A. Phthalic acid | 1. Hexane-1,6-dioic acid |
| B. Glutaric acid | 2. Benzene-1,2-dicarboxylic acid |
| C. Succinic acid | 3. Pentane-1,5-dioic acid |
| D. Adipic acid | 4. Butane-1,4-dioic acid |
Codes:
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 2 | 1 |
Which acid is least reactive to decarboxylation when heated?
| 1. |  |
2. |  |
| 3. |  |
4. |  |




