What is the ratio of the speed of an electron in the first orbit of an H-atom to the speed of light?
1.
2. 137
3.
4.
In the diagram shown below, two atomic transitions are shown. If then the value of λ will be:
1. 2000
2. 4000
3. 4500
4. 9000
Let f1 be the maximum frequency of the Lyman series, f2 be the frequency of the first line of the Lyman series, and f3 be the frequency of the series limit of the Balmer series, then which of the following is correct?
1. - =
2. - =
3. + =
4. 2 = +
If the wavelength of the first line in the Balmer Series of the hydrogen spectrum is λ, then what is the wavelength of the second line in this series?
1.
2.
3.
4.
In an atom, if the transition from n = 4 to n = 3 gives ultraviolet radiation, then to obtain infrared radiation, the transition should be:
| 1. | 5 → 4 | 2. | 3 → 2 |
| 3. | 2 → 1 | 4. | 3 → 1 |
Let R represent the orbital radius of an electron moving in an orbit and K represent its kinetic energy. Then the quantity KR varies with principal quantum number n as:
| 1. | 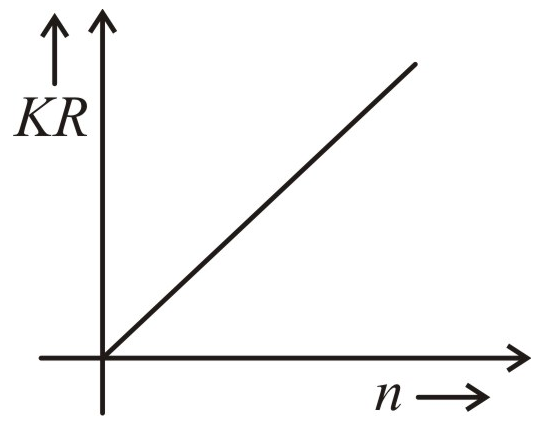 |
2. |  |
| 3. |  |
4. |  |
In an -particle scattering experiment, the number of particles scattered per minute in a direction perpendicular to the direction of incident particles is 40. What will be the number of particles scattered at an angle of 60° per minute?
| 1. | 145 | 2. | 160 |
| 3. | 172 | 4. | 157 |
The de-Broglie wavelength of an electron in the second orbit of a hydrogen atom is equal to:
| 1. | The perimeter of the orbit. |
| 2. | The half of the perimeter of the orbit. |
| 3. | The half of the diameter of the orbit. |
| 4. | The diameter of the orbit. |
What happens when an electron makes a transition from an excited state to the ground state of a hydrogen-like atom or ion?
| 1. | Its kinetic energy increases but potential energy and total energy decrease. |
| 2. | Kinetic energy, potential energy and total energy decrease. |
| 3. | Kinetic energy decreases, potential energy increases but the total energy remains the same. |
| 4. | Kinetic energy and total energy decrease but potential energy increases. |
A hydrogen atom is excited from the ground state to the state of principal quantum number 4. Then the number of spectral lines observed will be:
1. 3
2. 6
3. 5
4. 2



