The compound that is most difficult to protonate is:
| 1. |  |
2. | 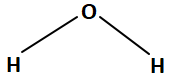 |
| 3. |  |
4. |  |
Subtopic: Acidic & Basic Character |
62%
From NCERT
NEET - 2019
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
What is the correct sequence of acidity for the following compounds?
| 1. | CH3COOH > BrCH2COOH > ClCH2COOH > FCH2COOH |
| 2. | FCH2COOH > CH3COOH > BrCH2COOH > ClCH2COOH |
| 3. | BrCH2COOH > ClCH2COOH > FCH2COOH > CH3COOH |
| 4. | FCH2COOH > ClCH2COOH > BrCH2COOH > CH3COOH |
Subtopic: Electron Displacement Effects | Acidic & Basic Character |
85%
From NCERT
AIPMT - 2007
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which one of the following compounds is most acidic:
| 1. |  |
2. |  |
| 3. |  |
4. | \(\mathrm{Cl}-\mathrm{CH}_2-\mathrm{CH}_2-\mathrm{OH}\) |
Subtopic: Acidic & Basic Character |
86%
From NCERT
AIPMT - 2005
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which of the following orders of acid strength is correct:
1. RCOOH > ROH > HOH > HC ≡ CH
2. RCOOH > HOH > ROH > HC ≡ CH
3. RCOOH > HOH > HC ≡ CH > ROH
4. RCOOH > HC ≡ CH > HOH > ROH
Subtopic: Acidic & Basic Character |
53%
From NCERT
AIPMT - 2003
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


