Select Chapter Topics:
The correct curve for Freundlich's adsorption isotherm is:
1.
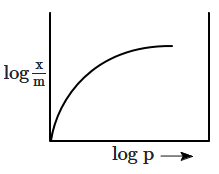
2.

3.

4.





Subtopic: Adsorption Isotherm |
64%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the types of colloidal systems given in Column I with the names given in Column II.
| Column I | Column II |
| A. Solid in Liquid | 1. Foam |
| B. Liquid in solid | 2. Sol |
| C. Liquid in Liquid | 3. Gel |
| D. Gas in Liquid | 4. Emulsion |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Emulsions |
90%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | An ordinary filter paper impregnated with collodion solution stops the flow of colloidal particles. |
| Reason (R): | The pore size of the filter paper becomes larger than the size of the colloidal particle. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Colloidal Solution |
60%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | Colloidal solutions do not show Brownian motion. |
| Reason (R): | Brownian motion is responsible for the stability of sols. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Colloidal Solution |
79%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | The coagulation power of Al3+ is more than Na+. |
| Reason (R): | Greater the valency of the flocculating ion added, the greater is its power to cause precipitation (Hardy-Schulze rule) |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Colloidal Solution |
90%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | Detergents with low CMC are more economical to use. |
| Reason (R): | The cleansing action of detergents involves the formation of micelles. These are formed when the concentration of detergents becomes equal to CMC. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Colloidal Solution |
62%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the items in Column I and Column II.
| Column I | Column II |
| A. Dialysis | 1. Cleansing action of soap |
| B. Peptization | 2. Coagulation |
| C. Emulsification | 3. Colloidal sol formation |
| D. Electrophoresis | 4. Purification |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 1 | 2 |
Subtopic: Colloidal Solution | Emulsions |
83%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The method of formation of the solution is given in Column I. Match it with the type of solution given in Column II.
| Column I | Column II |
| (A) Sulphur vapours passed through cold water | 1. Normal electrolyte solution |
| (B) Soap mixed with water above critical micelle concentration | 2. Molecular colloids |
| (C) White of egg whipped with water | 3. Associated colloid |
| (D) Soap mixed with water below critical micelle concentration | 4. Macromolecular colloids |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Colloidal Solution |
74%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the statement given in Column I with the phenomenon given in Column II.
| Column I | Column II |
| A. Dispersion medium moves in an electric field | 1. Osmosis |
| B. Solvent molecules pass through a semi-permeable membrane towards the solvent side | 2. Electrophoresis |
| C. Movement of charged colloidal particles under the influence of applied electric potential towards oppositely charged electrodes | 3. Electroosmosis |
| D. Solvent molecules pass through semi-permeable membranes towards the solution side | 4. Reverse osmosis |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 3 | 4 | 2 | 1 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Colloidal Solution |
68%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the items given in Column I and Column II.
| Column I | Column II |
| A. Protective colloid | 1. FeCl3 + NaOH |
| B. Liquid-liquid colloid | 2. Lyophilic colloids |
| C. Positively charged colloid | 3. Emulsion |
| D. Negatively charged colloid | 4. FeCl3 + hot water |
Codes
| A | B | C | D | |
| 1. | 3 | 2 | 4 | 1 |
| 2. | 1 | 4 | 3 | 2 |
| 3. | 2 | 3 | 4 | 1 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Emulsions |
89%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


