| Assertion (A): | For an isothermal physical process, the change in internal energy is equal to zero. |
| Reason (R): | The internal energy is directly proportional to temperature for physical processes. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyReason(R): All reactants and products are gases.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology| Assertion (A): | The increase in internal energy \((\Delta E ) \) for the vapourisation of one mole of water at 1 atm and 373 K is zero. |
| Reason (R): | For all isothermal processes, \(\Delta E = 0 \). |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True and (R) is False. |
| 4. | (A) and (R) both are False. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology| Assertion (A): | Absolute values of the internal energy of substances cannot be determined. |
| Reason (R): | It is impossible to determine the exact values of constituent energies of the substances. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyReason(R): During an isothermal process, the changes in the internal energy of a gas due to a decrease in pressure are nullified by the changes due to an increase in volume.
1. Both A and R are true and R is the correct explanation of A.
2. Both A and R are true but R is not the correct explanation of A.
3. A is true and R is false.
4. A and R both are false.
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology| Assertion (A): | ∆H is positive for endothermic reactions. |
| Reason (R): | If the total enthalpy of reactants and products are Hr & Hp respectively, then for an endothermic reaction HR < HP. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology| Assertion (A): | Cdiamond → Cgraphite ∆H and ∆U are the same for this reaction. |
| Reason (R): | Entropy increases during the conversion of diamond to graphite. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyFor one mole of an ideal gas, which of these statements must be true?
(I) U and H each depend only on temperature.
(II) Compressibility factor z is not equal to 1.
(III) CP, m – CV, m = R
(IV) dU = CVdT for any process.
1. (I), (III) and (IV)
2. (II), (III) and (IV)
3. (III) and (IV)
4. (I) and (III)
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
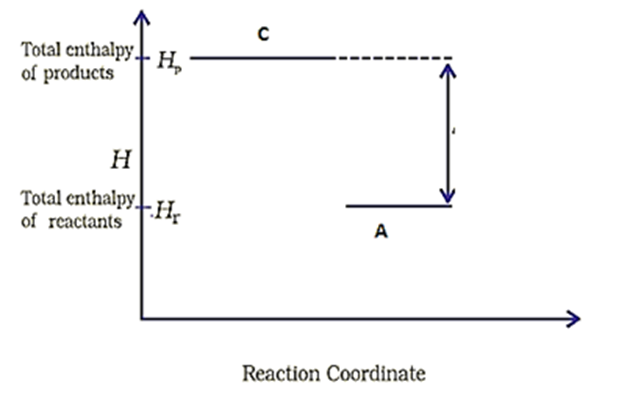
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyConsider the following diagram for a reaction .
The nature of the reaction is-
1. Exothermic
2. Endothermic
3. Reaction at equilibrium
4. None of the above
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyConsider the following diagram for a reaction
The nature of the reaction is-
| 1. | Exothermic | 2. | Endothermic |
| 3. | Reaction at equilibrium | 4. | None of the above |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology