Which graph represents the elevation in boiling point?
1.

2.

3.

4.
None of the above.



The following solutions were prepared by dissolving 10 g of glucose (C6H12O6) in 250 ml of water (P1), 10 g of urea (CH4N2O) in 250 ml of water (P2) and 10 g of sucrose (C12H22O11) in 250 ml of water (P3). The decreasing order of osmotic pressures of these solutions is:
| 1. | P2 > P3 > P1 | 2. | P3 > P1 > P2 |
| 3. | P2 > P1 > P3 | 4. | P1 > P2 > P3 |
The correct option for the value of vapour pressure of a solution at 45 C with benzene to octane in a molar ratio 3:2 is:
[At 45 C vapour pressure of benzene is 280 mm Hg and that of octane is 420 mm Hg. Assume Ideal gas]
| 1. | 336 mm of Hg | 2. | 350 mm of Hg |
| 3. | 160 mm of Hg | 4. | 168 mm of Hg |
Which graph represents the positive deviation of vapor pressure?
| 1. |  |
2. |  |
| 3. |  |
4. | None of the above |
The law depicted by the provided graph is:

| 1. | Henry's law | 2. | Raoult's law |
| 3. | Boyle's law | 4. | Charle's law |
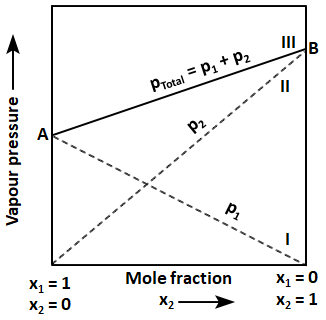
Points A and B in the below-mentioned graph represent, respectively:
| 1. | Partial pressures of first and second components |
| 2. | Vapour pressures of the pure second and first components |
| 3. | Partial pressures of second and first components |
| 4. | Vapour pressures of the pure first and second components |
The graph representing Henry's law is given below:

The value of the slope will be:
| 1. | \(4\times10^{-4}~\mathrm{torr}\) | 2. | \(5\times10^{-4}~\mathrm{torr}\) |
| 3. | \(4\times10^{4}~\mathrm{torr}\) | 4. | \(5\times10^{4}~\mathrm{torr}\) |
The type of molecular interactions in a solution of alcohol and water is:
| 1. | Electrostatic interaction | 2. | London dispersion forces |
| 3. | Hydrogen bonding | 4. | Ion-dipole interaction |
A drinking water sample was found to be contaminated with chloroform at a level of 15 ppm (by mass). What is the mass percent of chloroform in the sample?
1.
2.
3.
4.
An antifreeze solution is prepared from 222.6 g of ethylene glycol (C2H6O2) and 200 g of water. The molality of the solution would be:
| 1. | 6.9 m | 2. | 17.9 m |
| 3. | 29.9 m | 4. | 21.9 m |