2.15 What is the maximum number of emission lines when the excited electron of a H atom in n = 6 drops to the ground state?
When the excited electron of an H atom in n = 6 drops to the ground state, the following transitions are possible:
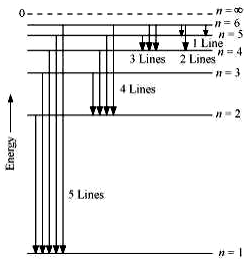
Hence, a total number of (5 + 4 + 3 + 2 + 1) 15 lines will be obtained in the emission spectrum.
The number of spectral lines produced when an electron in the n
th level drops down to the ground state is given by .
Given, n = 6
Number of spectral lines = = 15

© 2026 GoodEd Technologies Pvt. Ltd.