The mucosal layer in the stomach form irregular folds known as:-
1. villi
2. lumen
3. rugae
4. crypts of Lieberkuhn
The condition where urea accumulates in blood is:
1. Glycosuria
2. Uremia
3. Ketonuria
4. Acidosis
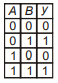
The trait shown in the given pedigree chart is most likely a/an:
1. Autosomal recessive trait
2. Autosomal dominant trait
3. Sex linked recessive trait
4. Sex linked dominant trait
Regulation of lac operon by repressor is referred to as:
1. Inducible regulation
2. Repressible regulation
3. Negative regulation
4. Positive regulation
What is true in the given figure?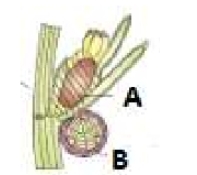
1. The plant concerned is Chara and A is Oogonium and B is Antheridium
2. The plant concerned is Chara and B is Oogonium and A is Antheridium
3. The plant concerned is Marchantia and A is Oogonium and b is Antheridium
4. The plant concerned is Marchantia and B is Oogonium
Match each item in Column I with one item in Column II regarding various classes of fungi and choose your answer from the codes given below:
|
Column I |
Column II |
||
|
I. |
Phycomycetes |
1. |
Sac fungi |
|
II. |
Ascomycetes |
2. |
Aseptate fungi |
|
III. |
Basidiomycetes |
3. |
Imperfect fungi |
|
IV. |
Deuteromycetes |
4. |
Puffballs |
Codes :
|
I |
II |
III |
IV |
|
|
1. |
1 |
2 |
3 |
4 |
|
2. |
2 |
1 |
4 |
3 |
|
3. |
2 |
1 |
3 |
4 |
|
4. |
1 |
2 |
4 |
3 |
Match each item in Column I with one item in Column II and choose your answer from the codes given below:
|
|
Column I |
|
Column II |
|
I. |
Potato spindle tuber disease |
1. |
Viroid |
|
II. |
Creutzfeldt Jakob disease |
2. |
Prion |
|
III. |
Mosaic disease of tobacco |
3. |
Virus |
|
IV. |
Rust of wheat |
4. |
Fungus |
Codes :
|
|
I |
II |
III |
IV |
|
1. |
1 |
2 |
3 |
4 |
|
2. |
2 |
1 |
4 |
3 |
|
3. |
2 |
1 |
3 |
4 |
|
4. |
1 |
2 |
4 |
3 |
Which of the following unicellular organism has a macronucleus for trophic function and one or more micronuclei for reproduction -
1. Trypanosoma
2. Paramecium
3. Euglena
4. Amoeba
Select correct match between Column I and II
| Column I | Column II | ||
| (a) | As quick referral system in taxonomical studies | (i) | Monograph |
| (b) | Collection of preserved plants and animals | (ii) | Herbaria |
| (c) | Index to the plant species found in a particular area | (iii) | Museum |
| (d) | Complete information of any one taxon | (iv) | Flora |
1. a-(iv), b-(iii), c-(ii), d-(i)
2. a-(ii), b-(iii), c-(i), d-(iv)
3. a-(iii), b-(ii), c-(iv), d-(i)
4. a-(ii), b-(iii), c-(iv), d-(i)
Flowering plants have developed many devices to discourage self pollination or to encourage cross pollination, except
1. Homogamy
2. Herkogamy
3. Dichogamy
4. Self incompatibility
Gene for starch synthesis in pea can show variable behaviour with respect to phenotype produced, it can show
1. Mendelian dominance
2. Multiple allelism
3. Incomplete dominance
4. More than one option is correct
Find odd one out with respect to autosomal recessive disorders
1. Cystic fibrosis
2. Myotonic dystrophy
3. Phenylketonuria
4. Sickle-cell anaemia
In eukaryotes, packaging of chromatin at higher level requires additional set of proteins called
1. Histones
2. NHC proteins
3. Polyamines
4. Basic proteins
The mRNA in eubacteria
1. Is polycistronic usually
2. Is processed before becoming functional
3. Has a cap at 5’ end
4. Has a tail at 3’ end
High aspartic acid, low nitrogen, and sugar content led to provide resistance against
1. Shoot borers in Okra
2. Jassids in Flat bean
3. Aphids in rapeseed
4. Stem borers in maize
The organism found as free living in rhizosphere and is an effective biocontrol agent of several plant pathogens is
1. Cercospora
2. Fusarium
3. Trichoderma
4. Bacillus
Find incorrect statement/s with respect to sewage treatment
(a) Primary treatment involves filtration and sedimentation of activated sludge
(b) ‘Flocs’ refers to masses of aerobic bacteria associated with fungal filaments to form mesh like structures
(c) Aerobic treatment continues till the BOD is reduced
(d) Aerobic bacteria kill other kind of bacteria and fungi present in the activated sludge
1. (b) & (d)
2. (a) & (c)
3. (b) & (c)
4. (a) & (d)
All given features are related to tropical rain forest biome, except
1. Maximum minerals stored in soil
2. Buttresses and leaf drip tips
3. Annual rainfall is 2000-3500 mm
4. Highly leached soil with low base content
Verhulst-Pearl logistic growth
(a) Occurs when resources are unlimited
(b) Is expressed as
(c) Exhibits sigmoid curve
(d) Is characterized by the function of carrying capacity
1. All are correct
2. Only (a) & (b) are correct
3. Only (a) is incorrect
4. Only (b) & (c) are incorrect
Plants of various successional stages of hydrosere are given, arrange them in order of their occurrence.
Nymphaea, Diatoms, Potamogeton, Cyperus, Typha and Populus
1. Diatoms, Potamogeton, Nymphaea, Typha, Cyperus, Populus
2. Diatoms, Nymphaea, Typha, Potamogeton, Cyperus, Populus
3. Diatoms, Typha, Nymphaea, Potamogeton, Populus, Cyperus
4. Diatoms, Potamogeton, Cyperus, Typha, Populus, Nymphaea
Most important factors to regulate the rate of decomposition are
1. Moisture and aeration
2. Chemical nature of detritus, soil pH
3. Aeration and soil pH
4. Temperature and moisture
Select an incorrect match (with respect to Protected Wild Life)
| 1. | Lion – Gir National Park |
| 2. | Tiger – Khangchenzonga National Park |
| 3. | One horned Rhino – Kaziranga National Park |
| 4. | Elephant – Manas Sanctuary |
(a) Western Ghats are more diverse than eastern ghats
(b) Valley’s are more diverse than mountains
(c) Tropics are more diverse than temperate areas
(d) Niche’s are narrow and more generalised in tropical areas
1. (a) & (d) are incorrect
2. Only (b) is correct
3. Only (d) is incorrect
4. Only (a) is incorrect
Find the correct match
| 1. | Montreal Protocol – 1992 |
| 2. | Kyoto Protocol – 1997 |
| 3. | Earth Summit – South Africa |
| 4. | World Summit – Brazil |
All given plants reproduce vegetatively using root buds, except
1. China rose
2. Dalbergia
3. Sweet potato
4. Asparagus
Litchi and pomegranate plants are commonly multiplied using
1. Air layering
2. Approach grafting
3. Bud grafting
4. Stem cuttings
Match the column-I with column-II
Column-I Column-II
a. Colletotrichum (i) Basidiomycetes
b. Agaricus (ii) Phycomycetes
c. Albugo (iii) Ascomycetes
d. Claviceps (iv) Deuteromycetes
1. a(i), b(iii), c(ii), d(iv)
2. a(ii), b(i), c(iii), d(iv)
3. a(iv), b(ii), c(i), d(iii)
4. a(iv), b(i), c(ii), d(iii)
Organisms belonging to chrysophyta shows
1. Non contractile vacuole
2. Presence of two flagella in grooves
3. Association with red tide
4. Siliceous frustules
Consider the given floral characters with respect to Fabaceae
(a) Zygomorphic flowers
(b) Vexillary calyx
(c) Marginal placentation
(d) Endospermic seed
(e) Diadelphous stamens
1. b, d & e are incorrect
2. a, c & e are correct
3. b, c & e are incorrect
4. a, b, c, d & e are correct
All given plants examplifies perigynous flowers, except
1. Guava
2. Plum
3. Rose
4. Peach
Banana, Pineapple and Chrysanthemum have some lateral branches arising for basal and underground portion of the main stem that grows obliquely out of the soil and gives rise to leafy shoots is called
1. Stolon
2. Runner
3. Offset
4. Sucker
Isobilateral leaf is different from dorsiventral leaf in
1. Having almost equal number of stomata on both the surfaces
2. Having undifferentiated mesophyll
3. Having bulliform cells in abaxial epidermal surface
4. More than one option is correct
Interfascicular cambium in a dicot stem is formed from
| 1. | Pericycle cells | 2. | Cortex cells |
| 3. | Medullary ray cells | 4. | Conjuctive parenchyma |
Identify the labels A, B and C in the following diagram
1. A-Central microtubule; B-Plasma membrane; C-Peripheral doublet
2. A-Central sheath; B-Plasma membrane; C-Peripheral doublet
3. A-Central sheath; B-Plasma membrane; C-Interdoublet bridge
4. A-Central microtubule; B-Peripheral doublet; C-Interdoublet bridge
Which organelle is extensive and continuous with the outer membrane of the nucleus?
1. Golgi complex
2. ER
3. Vacuole
4. Lysosome
The cells that do not divide further exit from cell cycle and enter an
| 1. | Inactive stage called G0 |
| 2. | Inactive stage called G2 |
| 3. | Active stage called G0 |
| 4. | Active stage called G1 |
Splitting of centromere occurs during
| 1. | Prophase-I and Anaphase I |
| 2. | Metaphase and Anaphase |
| 3. | Anaphase and Metaphase II |
| 4. | Anaphase and Anaphase II |
The water potential of pure water at standard temperatures, which is not under any pressure is taken to be equivalent to
1. Zero
2. –
3.
4. +
Reaction of N2 fixation process is depicted below
Select the correct set of A,B and C
1. A = , B=; C =
2. A = , B=; C =
3. A = , B=; C =
4. A = , B=; C =
Proton gradient develops across the thylakoid membrane due to all, except
| 1. | Splitting of water towards lumen side |
| 2. | NADP+ reduction towards stroma side |
| 3. | Movement of protons from lumen to stroma side by PQ |
| 4. | Release of H+ from stroma to lumen side by a H-carrier |
C4 and CAM plants have many similarities, like
| 1. | Secondary CO2 fixation by Rubisco |
| 2. | Scotoactive stomata |
| 3. | Primary CO2 fixation by PEPCase in bundle sheath |
| 4. | Presence of Kranz anatomy |
First action spectrum of photosynthesis was described by
1. Engelmann
2. Robert Hill
3. Sachs
4. Van Niel
Fate of pyruvate produced by glycolysis depends on cellular needs, it can enter
| 1. | Lactic acid fermentation | 2. | Alcoholic fermentation |
| 3. | Aerobic respiration | 4. | All are correct |
Select the correct match
| 1. | Auxin | – Promote seed dormancy |
| 2. | Cytokinin | – Overcoming apical dominance |
| 3. | Ethylene | – Bolting in beet |
| 4. | GA3 | – Thinning of cherry and walnut |
Genetic code is triplet and
1. Is ambiguous
2. Is read in contiguous fashion
3. Is non degenerate
4. Is non specific
The most distinctive feature of echinoderms is the presence of a water vascular system which helps in
(A) Locomotion
(B) Excretion
(C) Respiration
(D) Reproduction
1. (A) only
2. (B) & (C) only
3. (A), (B) & (C) only
4. (A), (B), (C) & (D)
Which of the following statement is incorrect with respect to vertebrates?
1. Vertebrates possess notochord during the embryonic period
2. Notochord is replaced by a cartilaginous or bony vertebral column in the adult
3. All vertebrates are chordates but all chordates are not vertebrates
4. Vertebrates have a dorsal muscular heart with two, three, or four chambers
The terrestrial animals who have crawling or creeping mode of locomotion and their body is covered by dry and
cornified skin are
(A) Calotes
(B) Testudo
(C) Hemidactylus
(D) Pterophyllum
1. A only
2. A & B only
3. A, B & C only
4. A, B, C & D
The junctions which facilitate the cells to communicate with each other by connecting the cytoplasm of adjoining cells for rapid transfer of ions, small molecules are
1. Tight junctions
2. Adhering junctions
3. Gap junctions
4. All of these
Which of the following is not an example of synovial joint?
1. Joint between atlas and axis
2. Joint between sternum and ribs
3. Joint between carpal and metacarpal of thumb
4. Joint between humerus and pectoral girdle
Scala vestibuli and scala tympani are linked with the middle ear at:
1. Oval window and round window, respectively
2. Round window and oval window, respectively
3. Oval window
4. Round window
Which of the following is incorrect with respect to parathyroid hormone (PTH)?
1. Secretion of PTH is regulated by circulating levels of calcium ions.
2. PTH acts on bones and stimulates the process of bone mineralisation.
3. PTH stimulates the reabsorption of Ca+2 by renal tubules.
4. PTH increases the absorption of Ca+2 from digested food.
A peptide hormone which causes dilation of blood vessels and decreases blood pressure is
1. Aldosterone
2. Adrenaline
3. Vasopressin
4. Atrial Natriuretic factor
ECG is a graphical representation of the electrical activity of the heart during a cardiac cycle in which the end of the T-wave marks:
1. End of ventricular systole
2. End of ventricular diastole
3. Beginning of ventricular systole
4. Both 2 & 3
Which of the following statement is correct with respect to to excretion?
1. Our lungs remove large amount of CO2 (180 litres/day) and also significant quantity of water everyday
2. The ascending limb is permeable to water and also allows transport of electrolytes actively or passively
3. Conditional reabsorption of Na+ and water takes place in proximal convoluted tubule
4. Maximum water reabsorption takes place in proximal convoluted tubule, even in the presence of ADH
Ability to abstract, reasoning, decision making, reality testing by judgement are regulated by
1. Frontal lobe
2. Parietal lobe
3. Temporal lobe
4. Occipital lobe
The disease caused due to degeneration of GABA secreting neurons of corpus striatum and acetylcholine secreting neurons of other part is
1. Paralysis agitans
2. Parkinson’s disease
3. Huntington’s chorea
4. Hodgkin’s disease
Long protein chain folded upon itself like a hollow woolen ball and gives us a 3-dimensional view of a protein belongs to
| 1. | Primary structure | 2. | Secondary structure |
| 3. | Tertiary structure | 4. | Quaternary structure |
In nucleic acid the bond between the phosphate and hydroxyl group of sugar is
1. Hydrogen bond
2. Glycosidic bond
3. Ester bond
4. Peptide bond
The maximum volume of air a person can breathe in after a forced expiration is
| (A) | Vital capacity | (B) | ERV + TV + IRV |
| (C) | TLC – RV | (D) | ERV – RV |
1. (A) only
2. (A) & (B) only
3. (A), (B) & (C) only
4. (A), (B), (C) & (D)
Which of the following cell completes the first meiotic division leading to the formation of two equal, haploid cells?
1. Spermatogonium
2. Primary spermatocytes
3. Secondary spermatocytes
4. Spermatids
The first sign of growing foetus may be noticed by
1. Development of limbs and digits
2. Listening to heart sound carefully through the stethoscope
3. Appearance of hair on the head
4. Formation of major organ systems
Match the column and choose the correct answer.
| Column-I | Column-II | ||
| a. | Hypothalamus | (i) | Sperm lysin |
| b. | Acrosome | (ii) | Estrogen |
| c. | Graffian follicle | (iii) | Relaxin |
| d. | Leydig cells | (iv) | GnRH |
| e. | Parturition | (v) | Testosterone |
1. a(iv), b(i), c(ii), d(iii), e(v)
2. a(ii), b(i), c(iv), d(iii), e(v)
3. a(ii), b(i), c(v), d(iv), e(iii)
4. a(iv), b(i), c(ii), d(v), e(iii)
Which of the following are ideal contraceptives for the female who wants to delay pregnancy and/or space children and one of the most widely accepted methods of contraception in India?
| 1. | Diaphragms |
| 2. | Intra uterine devices |
| 3. | Cervical caps |
| 4. | Oral contraceptives |
Which of the following will not show convergent evolution?
1. Eye of Octopus and mammal
2. Flippers of Penguins and Dolphins
3. Sweet potato and potato
4. Hearts of frog and rabbit
Which of the following factor will not affect Hardy-Weinberg equilibrium?
| 1. | Natural selection |
| 2. | Genetic drift |
| 3. | Genetic recombination |
| 4. | Random mating |
Proper burial of dead bodies for the first time started in which pre-historic man’s period?
1. Peking man
2. Java man
3. Neanderthal man
4. Cro-magnon man
Identify the wrong statements.
(A) Tumor of haemopoietic cells is called Leukemia
(B) Cancer arising from the epithelial tissues of internal organs and glands is referred as melanoma
(C) Sarcoma is a type of cancer where bone and cartilage are involved
(D) Only benign tumors are known as true cancer or neoplasm
1. (A) only
2. (B) & (C) only
3. (B) & (D) only
4. (A) & (B) only
Intestinal perforation and death may occur in severe cases of
1. Typhoid
2. Amoebiasis
3. Diarrhoea
4. Cholera
Which of the following statement is incorrect with respect to malaria?
1. Plasmodium falciparum is the most serious one and can even be fatal
2. Plasmodium enters the human body as sporozoites through the bite of infected female Anopheles
3. The rupture of RBCs is associated with release of a toxic substance, haemozoin
4. Parasites multiply within the mosquito body to form sporozoites and stored in the stomach of mosquito
Genetic engineering would not be possible if which of the following were not known?
1. Restriction endonuclease
2. DNA ligase
3. Reverse transcriptase
4. RNA synthetase
When the genotype of an organism is improved by the addition of foreign genes, the process is called
1. Biotechnology
2. Tissue culture
3. Genetic engineering
4. Genetic diversity
The product has to be subjected through a series of processes before it is ready for marketing as a finished product called down stream processing. It includes
1. Separation
2. Purification
3. Quality control
4. Both 1 & 2
Which of the following genes control the cotton bollworm?
1. cry IAc and cry IIAb
2. cry IIAb and cry IIAc
3. cry IAb and cry IAc
4. cry IAb and cry IIAc
The use of bio-resources by multinational companies and other organizations without proper authorization from the countries and people concerned without compensatory payment refers to
1. Biopiracy
2. Biosafety
3. Biopatency
4. All of these
The patient of diabetes mellitus is characterized by
1. Ketonuria
2. Glycosuria
3. Haematuria
4. Both 1 & 2
Select an incorrect statement regarding structural and functional features of histone proteins
1. They prevent transcription by bringing about supercoiling of DNA
2. They are rich in arginine and lysine
3. Their positively charged groups could interact with the negative charge of phosphate backbone of DNA
4. Five types of histones are associated with nu-body to form structural unit
Which of the following green house gas is produced from degradation of nitrogen fertilizers and denitrification in water logged soils?
1. CFC
2. N2O
3. CH4
4. CO2
Select incorrect statement with respect to logistic growth curve
1. Found in stable type of population
2. Lag phase is followed by exponential phase
3. A crash phase occurs at the end of growth
4. Population seldom grows beyond the carrying capacity of the area
Microtubular organelles in eukaryotes are essentially made up of which protein?
1. Flagellin
2. Dynein
3. Tubulin
4. Actin
Which of the following is both a green house gas as well as ozone depleting substance?
1. CO2
2. SO2
3. CFC
4. Water vapour
If in a population of 1000 individuals 60% individuals are heterozygous and 20% are homozygous dominants, then calculate the number of dominant alleles in population
1. 400
2. 1200
3. 1000
4. 1400
How many types of gametes will be produced by genotypes AaBbCcDDEe when all the genes are incompletely
linked?
1. 16
2. 8
3. 2
4. 4
On the basis of mode of secretion, mark the category of glands in which the secreting cells remain intact during the formation and discharge of secretion
1. Apocrine gland
2. Holocrine gland
3. Merocrine gland
4. Mixed gland
The synthesis of cDNA (complementary DNA) is carried out for which purpose?
1. To cut DNA at specific points, making small fragments
2. To make DNA copy of m-RNA
3. To join DNA fragments together
4. To separate fragments of DNA
Secretions of which glands in cockroach helps in the formation of ootheca?
1. Utricular gland
2. Conglobate gland
3. Phallic gland
4. Collaterial gland
Which of the following respiratory centre does not play role in basic respiratory rhythms but under enhanced respiratory drive, the respiratory signals of this centre fulfil the demand by regulating both inspiration and expiration?
1. Pneumotaxic centre
2. Apneustic centre
3. Dorsal respiratory group
4. Ventral respiratory group
In case the wife is unable to bear the child due to defective uterus, a surrogate mother can be hired for bearing the child, developed through the technique of
1. GIFT
2. ZIFT
3. IUI
4. IVF-ET
Untreated victims of SCID usually die from
1. Infections that in other normal people are minor
2. Anaphylactic shock
3. Congested lungs
4. Unusual high fever
Which of the following organ receives oxygenated blood only?
1. Liver
2. Lungs
3. Pancreas
4. Pituitary gland
Which of the alkali metal chloride is having the highest m.p. ?
1. LiCl
2. NaCl
3. KCl
4. RbCl
Sodium carbonate on heating gives:
1. water vapours
2. carbon dioxide
3. carbon dioxide + water vapour
4. none of the above
Which one of the following is likely to give a precipitate with solution?
1.
2.
3. (CH3)3
4.
The INCORRECT match among the following is:
1. \(\Delta G ^\circ < 0 , K_{eq} > 1\)
2. \(\Delta G ^\circ < 0 , K_{eq}< 1\)
3. \(\Delta G ^\circ = 0 , K_{eq} = 1\)
4. \(\Delta G ^\circ > 0 , K_{eq}< 1\)
Benzene diazonium chloride on reaction with aniline in the presence of dilute hydrochloric acid gives:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The INCORRECT statement is:
1. decomposes on heating to give .
2. Lithium is least reactive with water among the alkali metals.
3. LiCl crystallises from aqueous solution as .
4. Lithium is the strongest reducing agent among the alkali metals.
The at 298 K. The solubility (in mol/L) of in a 0.1 M solution is
1.
2.
3.
4.
The correct statement(s) about is(are)
(a) O–O bond lengths are equal.
(b) Thermal decomposition of is endothermic.
(c) is diamagnetic in nature.
(d) has a bent structure.
1. a,c,d
2. a,b,c
3.b,c,d
4,a,b,c,d
The primary pollutant that leads to photochemical smog is:
1. acrolein
2. nitrogen oxides
3. ozone
4. sulphur dioxide
The carbon–based reduction method is NOT used for the extraction of
1. tin from
2. iron from
3. aluminium from
4. lead from PbO
Thermal decomposition of a Mn compound (X) at 513 K results in compound Y, and a gaseous product. reacts with NaCl and concentrated to give a
pungent gas Z. X, Y and Z, respectively.
1.
2.
3.
4.
The molar solubility of CdM in water. The expected solubility of in a buffer solution of pH = 12 is :
1.
2.
3.
4.
P and Q are isomers of dicarboxylic acid . Both decolorize . On heating, P forms the cyclic anhydride.
Upon treatment with dilute alkaline KMnO4, P as well as Q could produce one or more than one from S, T and U.
Compounds formed from P and Q are, respectively
1. Optically active S and optically active pair (T, U)
2. Optically inactive S and optically active pair (T, U)
3. Optically active pair (T, U) and optically active S
4. Optically inactive pair (T, U) and optically inactive S
The compound used in the treatment of lead poisoning is:
1. EDTA
2. Cis-platin
3. D-penicillamine
4. desferrioxamine B
The pair that has similar atomic radii is:
1. Ti and Hf
2. Mn and Re
3. Sc and Ni
4. Mo and W
25 g of an unknown hydrocarbon upon burning produces 88 g of . This unknown hydrocarbon contains.
1. 24g of carbon and 1 g of hydrogen
2. 22g of carbon and 3 g of hydrogen
3. 18g of carbon and 7 g of hydrogen
4. 20g of carbon and 5 g of hydrogen
Match the chemical conversions in List – I with appropriate reagents in List – II and select the correct answer using the code given below the lists:
List-I List-II
(P) (i)
(Q) (ii) NaOEt
(R) (iii) Et-Br
(S) 
Codes:
P Q R S
(A) ii iii i iv
(B) iii ii i iv
(C) ii iii iv i
(D) iii ii iv i
For the identification of -naphthol using dye test, it is necessary to use
1. dichloromethane solution of -naphthol.
2. acidic solution of -naphthol.
3. neutral solution of -naphthol.
4. alkaline solution of -naphthol
The major product in the following reaction is
Under ambient conditions, the total number of gases released as products in the final step of the reaction scheme shown below is
1. 0
2. 1
3. 2
4. 3
required for a reaction is produced by the decomposition of as per the equation
The initial concentration of is 3.00 mol and it is 2.75 mol after 30 minutes. The rate of formation of is
1.
2.
3.
4.
The correct name of the following polymer is: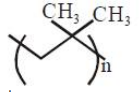
1. Polyisoprene
2. Polytert-butylene
3. Polyisobutane
4. Polyisobutylene
Heating of 2-chloro-1-phenylbutane with EtOK/EtOH gives X as the major product. Reaction of X with Hg followed by gives Y as the major product. Y is:
1. 
2. 
3. 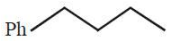
4. 
The IUPAC name of the following compound is :
1. 3, 5-dimethyl-4-propylhept-1-en-6-yne
2. 3-methyl-4-(1-methylprop-2-ynyl)-1-heptene
3. 3-methyl-4-(3-methylprop-1-enyl)-1-heptyne
4. 3, 5-dimethyl-4-propylhept-6-en-1-yne
In comparison to boron, berylium has:
1. greater nuclear charge and greater first ionisation enthalpy
2. lesser nuclear charge and lesser first ionisation enthalpy
3. greater nuclear charge and lesser first ionisation enthalpy
4. lesser nuclear charge and greater first ionisation enthalpy
A solution is prepared by dissolving 0.6 g of urea (molar mass = 60 g ) and 1.8 g of glucose (molar mass = 180 g ) in 100 mL of water at . The osmotic pressure of the solution is :
1. 8.2 atm
2. 1.64 atm
3. 4.92 atm
4. 2.46 atm
Hydrogen peroxide in its reaction with and respectively, is acting as a
1. reducing agent, oxidising agent
2. reducing agent, reducing agent
3. oxidising agent, oxidising agent
4. oxidising agent, reducing agent
The acidic hydrolysis of ether (X) shown below is fastest when
1. one phenyl group is replaced by a methyl group.
2. one phenyl group is replaced by a para-methoxyphenyl group.
3. two phenyl groups are replaced by two para-methoxyphenyl groups.
4. no structural change is made to X.
X and Y are two volatile liquids with molar weights of 10 g and 40 g respectively. Two cotton plugs, one soaked in X and the other soaked in Y, are simultaneously placed at the ends of a tube of length L = 24 cm, as shown in the figure. The tube is filled with an inert gas at 1 atmosphere pressure and a temperature of 300 K. Vapours of X and Y react to form a product which is first observed at a distance d cm from the plug soaked in X.
Take X and Y to have equal molecular diameters and assume ideal behaviour for the inert gas and the two vapours.
The value of d in cm (shown in the figure), as estimated from Graham’s law, is
1. 8
2. 12
3. 16
4. 20
Match the orbital overlap figures shown in List-I with the description given in List-II and select the correct answer using the code given below the lists.
List-I List-II
P. 
Q. 
R. 
S. 
codes:
P Q R S
1. ii i iii iv
2. iv iii i ii
3. ii iii i iv
4. iv i iii ii
The species that can have a trans-isomer is:
(en = ethane-1,2-diamine, ox = oxalate)
1.
2. [Pt(en)2Cl2]2+
3. [Cr(en)2(ox)]+
4.
Which of the following orbital has the graph between and r(radial distance) given below:
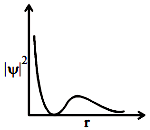
1. 1s orbital
2. 3s orbital
3. 2s orbital
4. 2p orbital
| 1. | II > I > III | 2. | III > II > I |
| 3. | I > II > III | 4. | III > I > II |
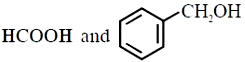
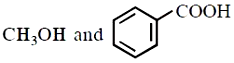
Major products 'P' of the following reaction are:

1. \(\mathrm{CH}_3 \mathrm{OH} \text { and } \mathrm{HCO}_2 \mathrm{H}\)
2. 
3. 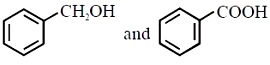
4. 
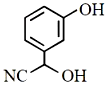
The major product of the following reaction is: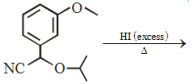
1.  2.
2. 
3. 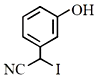 4.
4. 
Match the refining methods (Column I) with metals (Column II).
Column I Column II
(Refining methods) (Metals)
(I) Liquation (a) Zr
(II) zone Refining (b) Ni
(III) Mond process (C) Sn
(IV) Van Arkel Method (d) Ga
1. (I) – (b); (II) – (c); (III) – (d); (IV) – (a)
2. (I) – (b); (II) – (d); (III) – (a); (IV) – (c)
3. (I) – (c); (II) – (a); (III) – (b); (IV) – (d)
4. (I) – (c); (II) – (d); (III) – (b); (IV) – (a)
Ethylamine (C2H5NH2) can be obtained from N-ethylphthalimide on treatment with:
1.
2. /H+
3.
4.
The correct order of catenation is:
1. C > Sn > Si Ge
2. Ge > Sn > Si > C
3. Si > Sn > C > Ge
4. C > Si > Ge Sn
The major product of the following reaction is

1. 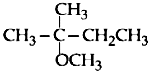
2. 
3. 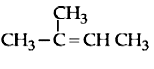
4. 
The oxoacid of sulfur that does not contain a bond between sulfur atoms is:
1.
2.
3.
4.
The alloy used in the construction of aircraft is
1. Mg–Mn
2. Mg-Zn
3. Mg-Al
4. Mg-Sn
At room temperature, a dilute solution of urea is prepared by dissolving 0.60 g of urea in 360 g of water. If the vapor pressure of pure water at this temperature is 35 mm Hg, lowering of vapor pressure will be: (molar mass of urea = 60 g )
1. 0.027 mmHg
2. 0.031 mmHg
3. 0.028 mmHg
4. 0.017 mmHg
Determine the kinetic energy of an electron located in the second energy level of a hydrogen atom according to the Bohr model, with a0 representing the Bohr radius.
1.
2.
3.
4.
For one mole of a van der Waals gas when b = 0 and T = 300 K, the PV vs. 1/V plot is shown below. The value of the van der Waals constant a is
1. 1.0
2. 4.5
3. 1.5
4. 3.0
29.2 % (w/w) HCl stock solution has density of 1.25 . The molecular weight of HCl is 36.5 g . The volume (mL) of stock solution required to prepare a 200 mL solution of 0.4 M HCl is
1. 20 ml
2. 100 ml
3. 36 ml
4. 8 ml
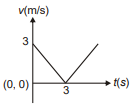
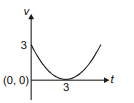
Position of a particle moving along the y-axis is given by , then which of the following represent the correct variation of speed (v) with respect to time (t)?
1. 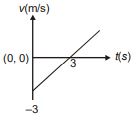 2.
2. 
3. 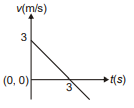 4.
4. 
The sides of a rectangle are measured to be \(2.5\) cm and \(15.50\) cm. The area of the rectangle will be:
1. \(3.9~\text{cm}^2\)
2. \(38.8~\text{cm}^2\)
3. \(0.39 \times10^2~\text{cm}^2\)
4. \(38.75~\text{cm}^2\)
The pressure P of a real gas is given as , where symbols have their usual meanings. The dimensions of is same as-
1. Torque
2. Stress
3. Strain
4. Coefficient of viscocity
If a particle starts from rest along a straight line, with constant acceleration , then its average speed during journey in which it travels a distance d from starting point is given by
1.
2.
3.
4.
A particle is projected from the ground at an angle from horizontal with an initial velocity u. At height 45 cm from the ground, the velocity of the projectile is 4 m/s horizontally. The angle of projection with horizontal is (g = 10 )
1. 45°
2. 53°
3. 37°
4. 60°
A man moving in the west direction observes wind is blowing towards the south. If the man doubles his speed in the same direction, then the direction of wind with respect to man will be:
1. North-West
2. South-West
3. North-East
4. East-South
Two blocks of masses = 12 kg are connected by a light string passing over a light frictionless pulley as shown. If the system is in equilibrium, then force exerted by inclined plane on the block is

1. 60 N
2. 60N
3. 120 N
4. 120N
Two blocks of masses m and 2m are connected by a light spring of spring constant K and are kept on a horizontal smooth surface. The blocks are moved far apart so that spring is elongated by x unit and then released. The relative acceleration of one block with respect to other just after the release is
1.
2.
3.
4.
Two identical thin rods are used to form a cross as shown. Each rod has mass m and length l. The moment of inertia of the system of rods about a Z-axis is

1.
2.
3.
4.
If the gravitational potential energy of a body at a distance R from the center of the earth is U, then the weight of the body at a distance R from the surface of the earth is numerically equal to (R = Radius of the earth).
1.
2.
3.
4.
Two small satellites are orbiting the earth in circular orbits of radii r and 2r respectively. If their masses are in the ratio of 2:1, then the ratio of their time periods of revolution about the earth will be
1. 1: 1
2.
3. 1:
4.
Kepler's law of planetary motion is based on the law of conservation of
1. Momentum
2. Kinetic energy
3. Angular momentum
4. Angular velocity
Select correct statement(s) regarding transistors.
1. Collector current is almost independent of collector-emitter voltage (in common emitter configuration) in the active mode
2. Transistor is used as a switch in active mode
3. Transistor is used as an amplifier in saturation mode
4. Collector part of the transistor is heavily doped
1 g ice at 0°C and 1 g steam at 100°C are mixed together. Assuming no heat loss, then the final temperature of the mixture will be
1. 25°C
2. 35°C
3. 85°C
4. 100°C
Which of the following statement is correct regarding heat transfer?
1. Forced convection requires gravity
2. Natural convection requires gravity
3. Radiation and forced convection requires gravity
4. Neither natural nor forced convection requires gravity
If 200 J of heat is added to a system and system does work equal to 250 J, then the change in the internal energy of the system is
1. 50 J
2. 450 J
3. -50 J
4. -450 J
An ideal gas undergoes a cyclic process ABCA as shown.
The heat exchange between the system and the surrounding during the process will be:
| 1. | 10 J | 2. | 5 J |
| 3. | 15 J | 4. | 20 J |
Linear momentum of a body is increased by 50% keeping mass constant. Percentage increase in its kinetic energy is
1. 50%
2. 100%
3. 125%
4. 75%
A block A is placed on a horizontal platform B which undergoes S.H.M. about mean position as shown. The block A does not slip on platform B, then the force of friction between block A and platform B is

1. Always directed towards mean position
2. Directed towards mean when the block is moving away from mean position and it is directed away from mean when the block is moving towards mean position
3. Directed towards left irrespective of the position of block A
4. Directed towards right irrespective of the position of block A
An organ pipe filled with a gas at 27°C resonates at 400 Hz in its fundamental mode. If it is filled with the same gas at temperature 90°C, then the resonance frequency will be
1. 450 Hz
2. 440 Hz
3. 512 Hz
4. 480 Hz
If the frequency of the stationary sound source appears to drop by 10% to a moving person, then the velocity of the person is (velocity of sound is 330 m/s and motion of a person is along the line connecting source and person)
(1) 22 m/s
(2) 33 m/s
(3) 44 m/s
(4) 55 m/s
When two coherent waves each of intensity superpose, then the maximum possible intensity is
1.
2. 2
3. 3
4. 4
A charge is kept at distance \(r(r>R)\) from the center of an uncharged conducting hollow sphere as shown. The electric potential at its center due to induced
charges is:
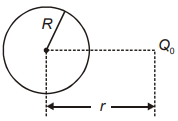
1. \(\frac{Q_0}{4\pi\epsilon ~r}\)
2. \(\frac{Q_0}{4\pi\epsilon ~R}\)
3. \(\frac{Q_0}{4\pi\epsilon ~(R+r)}\)
4. Zero
What is the ratio of current drawn from the battery at t = 0 and after a long time of closing the key in the following circuit?
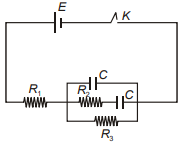
1.
2.
3.
4.
Two ideal diodes and two capacitors of capacitance C and 2C are connected as shown. What is the output voltage between the terminals a and b?
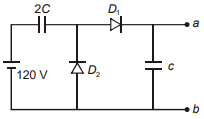
1. 80V
2. 40V
3. 60V
4. 120V
The equivalent capacitance between the points \(A\) and \(B\) in the given network is:

1. \(25~\mu\text{F}\)
2. \(16~\mu\text{F}\)
3. \(21~\mu\text{F}\)
4. \(12~\mu\text{F}\)
The temperature coefficient of resistance for a semiconductor is
1. Positive
2. Negative
3. Infinite
4. Zero
What is the potential difference between points A and B in the given network?

1. 2V
2. 3V
3. 6V
4. 5V
A battery of 10 volt and internal resistance 30 is connected to a variable resistance R. The power dissipated in R is maximum when R is equal to -
1. 30
2. 15
3. 60
4. 20
A cell of emf E and internal resistance r is connected in series with an external resistance nr. The ratio of the terminal potential to emf of the cell is
1.
2.
3.
4. n
The mass of two Cu wires is in the ratio of 1: 2 and their lengths are in the ratio of 3: 2. The ratio of their resistances are
1. 5: 1
2. 3: 1
3. 4: 1
4. 9: 2
An infinitely long conductor is kept near a square conducting loop of side 'a' as shown. If the current in the long wire increases with time then, current induced in the loop is

1. Zero
2. First clockwise then anticlockwise
3. Clockwise
4. Anticlockwise
A rod \(AB\) of length \(l\) is moving with constant speed \(v\) in a uniform magnetic field on a conducting \(U\)-shaped wire as shown. If the rate of loss of heat energy across resistance \(R\) is \(Q,\) then the force needed parallel to velocity to keep rod moving with constant speed \(v\) is:

1. \(Qv\)
2. \(\dfrac{Q}{v}\)
3. \(\dfrac{Q^2}{v}\)
4. \(Q^2v\)
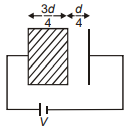
A dielectric of dielectric constant K = 3 is kept between the two plates of parallel plate capacitor such that it occupies of space between the plates as shown. What percentage of energy is stored inside dielectric material?
1. 25%
2. 30%
3. 50%
4. 60%
Select the incorrect statement regarding magnetic material.
| 1. | The magnetic dipole moment of each atom of para and ferromagnetic substance is zero and that of diamagnetic material is non-zero in the absence of external magnetizing force |
| 2. | When the diamagnetic substance is brought near the north or south pole of a bar magnet, it is repelled |
| 3. | Magnetic susceptibility of diamagnetic substances is temperature independent and that of para and ferromagnetic substances is temperature-dependent |
| 4. | All of these |
A closed-loop ABCD carrying current is kept in a region having a uniform magnetic field, such that the sides AB, BC, CD, and DA experience force respectively which are in the plane of the loop. The force on the side DA will be

1.
2.
3.
4.
The focal length of the glass piece shown in the figure is (Both surfaces have an equal radius R).

1.
2. Infinity
3.
4.
A ray of monochromatic light is incident normally on one refracting surface of the prism of angle 30°. If the refractive index of prism material is , then the deviation produced by the prism is
1. 15°
2. 30°
3. 60°
4. 90°
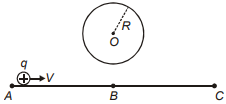
A proton is moving along a straight line ABC as shown. The direction of induced current in the conducting circular loop is
1. Clockwise
2. Anticlockwise
3. First clockwise and then anticlockwise
4. First anticlockwise and then clockwise
Four current carrying circular loops are kept in the uniform magnetic field in different orientations as shown. Arrange them in decreasing order of potential energy.
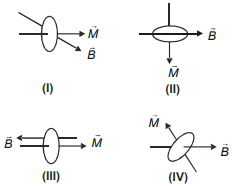
1. IV > III > I > II
2. III > IV > I > II
3. II > I > III > IV
4. III > IV > II > I
A beam of light falls normally on a plane surface. This surface reflects 40% of incident light and the rest is absorbed. If the power of the incident beam is 24 W, then the force exerted by the beam on the surface is
1. 1.12 × N
2. 1.12 × N
3. 1.12 × N
4. 1.12 × N
When the frequency of incident radiation on a metal surface is v, the maximum KE of the photoelectron is . If the frequency of incident radiation is doubled, then the maximum KE becomes K. Select correct statement(s).
1. K = 2
2. K < 2
3. K =
4. K > 2
When the plane surface of a plano-convex lens is silvered, it behaves like a concave mirror of focal length 30 cm. The radius of curvature of the convex surface is - (refractive index of the lens material is )
1. 20 cm
2. 30 cm
3. 15 cm
4. 25 cm
The potential difference across the resistor, the capacitor and inductor of series LCR circuit are 50 V, 100 V, and 100 V respectively. Power factor of the circuit is
1. Zero
2. 1
3.
4.
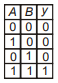
Which of the following truth table is correct for function y in the given combination of logic gates?
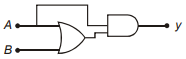
1. 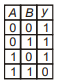 2.
2. 
3. 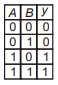 4.
4.