Which one of the following is important as one of the first line of defence against inhaled and ingested pathogen ?
1. IgA
2. IgG
3. IgM
4. IgD
A person suffering from a disease caused by Plasmodium, experiences chill and fever at the time when?
| 1. | The sporozites released from RBCs are being rapidly killed and broken down inside spleen |
| 2. | The trophozites reach maximum growth and give out certain toxins |
| 3. | The parasite after its rapid multiplication inside RBCs ruptures them, releasing the stage to enter fresh RBCs |
| 4. | The microgametocytes and megagametocytes are being destroyed by the WBCs |
A person who met with a road accident is likely to develop tetanus, can be immunised by administering
1. Weakened germs
2. Dead germs
3. Preformed antibodies
4. Wide spectrum antibiotics
MALT constitutes about ______ percent of the lymphoid tissue in human body
1. 50%
2. 20%
3. 70 %
4. 10%
Acid in stomach, saliva in the mouth, tears from eyes, all prevent microbial growth belong to which of the following barriers?
1. Physical barrier
2. Physiological barrier
3. Cellular barrier
4. Cytokine barrier
Which of the following is wrongly matched in the given table ?
| Microbe | Product | Application | |
| 1. |
Clostridium |
Lipase |
Removal of |
| 2. |
Trichoderma |
Cyclosporin A |
Immunosuppressive |
| 3. |
Monascuspur |
Statins |
Lowering of |
| 4. | Streptococcus | Streptokinase |
Removal of clot |
During sewage treatment, biogases are produced which include :
1. Methane,oxygen, hydrogen sulphide
2. Hydrogen sulphide, methane, sulphur dioxide
3. Methane, hydrogen sulphide, carbon dioxide
4. Hydrogen sulphide, nitrogen, methane
All are correct with respect to BOD (Biochemical oxygen Demand), except
1. It refers to the amount of oxygen that would be consumed if all the organic matter in one liter of water were oxidized by bacteria
2. The BOD test is a measure of the organic matter present in the water
3. The greater the BOD of waste water, less is its polluting potential
4. Waste water is treated till BOD is reduced significantly
What are flocs?
(1) Masses of anaerobic bacteria
(2) Masses of aerobic fungi only
(3) Masses of anaerobic bacteria and fungi
(4) Masses of aerobic bacteria associated with fungal filaments
Select the correct statement from the following
(1) Activated sludge-sediment in settlement tanks of sewage treatment plant is a rich source of aerobic bacteria
(2) Biogas is produced by the activity of aerobic bacteria on animal waste
(3) Methanobacterium is an aerobic bacterium found in rumen of cattle
(4) Biogas, commonly called gobar gas is pure methane
One of the major difficulties in the biological control of insect/pest is that
(1) The method is less effective as compared with the use of insecticides
(2) The practical difficulty of introducing the predator to specific areas
(3) The predator develops a preference to other deits and may itself become a pest
(4) The predator does not always survive when transferred to a new environment
For the stability of the organic compounds, Oparin opined that probably:
1. UV radiation never reached the surface of the primeval Earth
2. the primitive Earth was in a molten state
3. the primitive Earth atmosphere was reducing rather than oxidizing
4. plenty of energy sources were available for chemical reactions to occur
Adenine was synthesized by Orgel, using the Miller apparatus, when he used the gas :
1. carbon dioxide
2. hydrogen cyanide
3. hydrogen sulfide
4. nitrogen
The first genetic code of life was based on :
1. DNA
2. RNA
3. Proteins
4. Lipids
Both Charles Darwin and A.R.Wallace were influenced by the:
1. writings of J.B.S.Haldane on the origin of life
2. findings of Urey and Miller experiments
3. findings on the Galapagos Islands
4. essay on human population by T.R.Malthus
What is the ultimate source of genetic variability?
1. mutation
2. migration
3. genetic drift
4. nonrandom mating
de Vries based his “Mutation Theory” on his observation on :
1. Pisum sativum
2. Drosophila melanogaster
3. Oenothera lamackiana
4. Coenorhabditis elegans
According to the modern view of Darwinism, the unit of natural selection is the :
1. population
2. individual
3. species
4. community
If there are two forms of a specific enzyme in a population, and if the relative frequencies of the individuals possessing each form of the enzyme changes, then certainly there is :
1. mutation
2. genetic drift
3. gene migration
4. evolution
The cause of the high incidence of the allele of sickle cell anaemia in human population is due to :
1. genetic drift
2. natural selection
3. bottleneck effect
4. founder effect
Which of the following is the most important precondition for speciation to occur?
1. reproductive isolation
2. a crash of the population size
3. adaptive radiation
4. random mating
Which of the following pairs of organs do not represent analogous organs?
1. Eye of an octopus and eye of a mammal
2. Flippers of penguin and flippers of dolphin
3. Wings of bat and forelimbs of humans
4. Wings of birds and wings of butterfly
The only evolutionary agent that produces adaptive evolutionary changes is :
1. Non random mating
2. Mutation
3. Genetic drift
4. Natural selection
A population of an animal is hunted down to only 20 organisms. Later the number is restored to 10,000 organisms, but the variations in this population are highly reduced. This is called :
1. Founder effect
2. Bottleneck effect
3. Edge effect
4. Heterosis
A change in allele frequency within a population, over a succession of generations , is called :
1. microevolution, or adaptive evolution
2. macroevolution, or speciation
3. coevolution
4. phylogenetic evolution
What key characteristic of T2 bacteriophage allowed Hershey and Chase to use it in their studies of the genetic material?
| 1. | Its genes encode proteins that assemble to produce the viral coat. |
| 2. | It injects its genetic material into a bacterial cell. |
| 3. | It can undergo either the lytic or lysogenic life cycle. |
| 4. | It enters the bacterial cell to cause infection. |
Which of the following statements about the structure of DNA is incorrect?
1. One complete turn requires 3.4 nm and 10 base pairs.
2. The backbones of each strand run in opposite directions relative to each other.
3. Each pair of nucleotides is held together by three hydrogen bonds.
4. The width of the molecule is a constant 2nm.
Which of the following structures of a eukaryotic chromosome is not primarily composed of DNA?
1. Telomeres.
2. Origin of replication.
3. Kinetochore.
4. Centromere.
Which statement about bacterial DNA replication is correct?
1. DNA replication begins at several places along the chromosome.
2. DNA replication begins at the origin and travels in both directions.
3. DNA replication begins at the origin and travels around the chromosome back to the origin.
4. DNA replication begins at a GC rich region of the chromosome.
Which statement could NOT describe both eukaryotic and prokaryotic transcription?
1. Promoter elements in the DNA are required for polymerase binding.
2. Transcription factors control the binding of RNA polymerase.
3. RNA polymerase opens a double stranded DNA to expose the template strand.
4. RNA polymerase catalyzes RNA synthesis from 3' to 5' of the new strand
Which of the following would you expect to find in an inducible system?
1. A repressor protein, which is bound to DNA in absence of any other factor.
2. A repressor protein, which is bound to DNA in the presence of a corepressor.
3. An activator protein, which is bound to DNA in absence of any other factor.
4. An activator protein, which is bound to DNA only in the absence of an inhibitor.
A genetic cross between homozygous individuals but with different alleles for a single gene of interest is called as:
1. A reciprocal cross
2. Monohybrid cross
3. Dihybrid cross
4. Test cross
In a test cross, the plant expressing the dominant phenotype is crossed with the plant:
1. Which is heterozygous dominant
2. Which is heterozygous recessive
3. Which is homozygous recessive
4. Which is homozygous dominant
Which of the following is not true for the Law of Dominance given by Mendel?
| 1. | Characters are controlled by discrete units called factors. |
| 2. | Factors occur in pairs. |
| 3. | In a dissimilar pair of factors one member of the pair dominates the other. |
| 4. | The alleles show blending at the time of fertilization |
A heterozygous plant produces:
1. Only one kind of gamete
2. Two kinds of gametes each having one allele with equal proportion.
3. Two kinds of gametes each having two alleles with equal proportion.
4. Two kinds of gametes each having one allele with one allele occurring in more gametes than the other.
What is the F2 phenotypic ratio in cases of incomplete dominance?
1. 1 : 1
2. 3 : 1
3. 1 : 2 : 1
4. 1 : 1
Blood group antigens are:
1. Carbohydrates present in plasma
2. Carbohydrates present on the surface of RBCs
3. Plasma proteins
4. Proteins present on the surface of RBCs
What can be the blood groups of progeny whose father and mother are of AB and O blood group respectively?
1. A and B only
2. AB only
3. All except O
4. A, B, AB and O
Occasionally, a single gene product may produce more than one effect. Such a gene is said to be:
1. Pleiotropic
2. Polygenic
3. Psuedoallele
4. Housekeeping
‘When two pairs of traits are combined in a hybrid, segregation of one pair of characters is independent of the other pair of characters’. This is the statement of:
1. Law of Dominance
2. Law of segregation
3. Law of Independent Assortment
4. Law of Linkage
Mendel’s work remained unrecognized till 1900. Which of the following was not a reason for this?
1. His work was widely publicized and it brought bad name to Mendel
2. His concept of factors as stable and discrete units that did not ‘blend’ was not accepted.
3. His approach of using mathematics to explain biological phenomena was unacceptable.
4. He could not provide any physical proof for the existence of factors or say what they were made of.
“Chromosomes, which are seen in all dividing cells and pass from one generation to the next, are the basis for all genetic inheritance”.
This statement is credited to:
1. Watson and Crick
2. Hershey and Chase
3. Sutton and Boveri
4. Meselson and Stahl
When the two genes in a dihybrid cross are situated on the same chromosome:
1. The proportion of parental gene combinations was much higher than the non-parental type.
2. The proportion of parental gene combinations was much lesser than the non-parental type.
3. The proportion of parental gene combinations was equal to the non-parental type.
4. Only recombinants are formed.
The mechanism of sex determination in grasshoppers is:
1. XX – XY; male heterogamety
2. XX – XY; female heterogamety
3. XX – XO; male heterogamety
4. XX – XO; female heterogamety
During his observation of spermatogenesis in a few insects, Henking found that a nuclear structure was received by 50 % of the sperms. What did he call this structure?
1. X – body
2. Y – body
3. X – chromosome
4. Y – chromosome
It is unfortunate that in our society women are blamed for producing female children and have been ostracised and ill-treated because:
1. The sex is determined by the type of sperm fertilizing the egg
2. The sex is determined by the type of egg fertilizing the sperm
3. The sex is determined by the hormones produced by the fetus
4. The sex is determined by God’s Will
Mendelian disorders are mainly determined by:
1. Alteration or mutation in a single gene.
2. Chromosomal gross structural changes.
3. Recombination between linked genes.
4. Jumping genes
The family pedigree of Queen Victoria shows a number of haemophilic descendants as she was:
1. Affected by the disease
2. Carrier for the disease
3. Did not carry the allele for haemophilia
4. Was not a queen
The point mutation leading to sickle cell anaemia occurs:
1. at sixth position in the beta chain of haemoglobin
2. at sixth position in the alpha chain of haemoglobin
3. at sixth position in the beta chain of myoglobin
4. at sixth position in the alpha chain of myoglobin
In cases of Phenylketonuria:
1. The affected individual lacks an enzyme that converts the amino acid tyrosine into phenylalanine.
2. The affected individual makes an enzyme that converts the amino acid tyrosine into phenylalanine.
3. The affected individual makes an enzyme that converts the amino acid phenylalanine into tyrosine.
4. The affected individual lacks an enzyme that converts the amino acid phenylalanine into tyrosine.
Monosomy means:
1. Lack of one pair of chromosomes
2. Lack of one of any one pair of chromosome
3. Presence of an extra chromosome
4. Presence of an extra set of chromosomes
According to Erwin Chargaff, for a double stranded DNA
1. The ratios between Adenine and Thymine, and , Guanine and Cytosine are constant and equals one.
2. The ratios between Adenine and Thymine, and , Guanine and Cytosine are constant but is not equal to one.
3. The ratios between Adenine and Guanine, and , Thymine and Cytosine are constant and equals one.
4. The ratios between Adenine and Guanine, and , Thymine and Cytosine are constant but is not equal to one.
A molecule that can act as a genetic material must fulfill all the following criteria except:
1. It should be able to generate its replica
2. It should chemically and structurally be stable
3. It should provide scope for rapid mutations
4. It should be able to express itself in the form of “Mendelian Characters”
Reverse transcriptase is a:
1. DNA dependent RNA polymerase
2. RNA dependent DNA polymerase
3. RNA dependent RNA polymerase
4. DNA dependent DNA polymerase
DNA replication is correctly described as:
1. Semi-conservative and Continuous
2. Semi-conservative and Discontinuous
3. Conservative and Continuous
4. Semi-conservative and Semi-discontinuous
In a transcription unit, with respect to structural gene, the promoter is located:
1. Upstream and 5’
2. Upstream and 3’
3. Downstream and 5’
4. Downstream and 3’
In most prokaryotes, the transcription unit is:
1. Mono-cistronic
2. Poly-cistronic
3. Multi-cistronic
4. Uni-cistronic
Whose cell free system finally helped the genetic code to be deciphered?
1. Gamow
2. Severo Ochoa
3. Hargobind Khorana
4. Marshall Nirenberg
Which of the following is not a stop codon?
1. UAA
2. UGA
3. UAC
4. UAG
Which of the following anticodons will hybridize with the mRNA codon 5’ – AUG – 3’?
1. 5’ – UAC – 3’
2. 5’ – TAC – 3’
3. 3’ – UAC – 5’
4. 3’ – TAC – 5’
In Australia, marsupials and placental mammals have evolved to share many similar characteristics. This type of evolution may be referred to as:
| 1. | Adaptive Radiation | 2. | Divergent Evolution |
| 3. | Cyclical Evolution | 4. | Convergent Evolution |
| Column-I | Column-II | ||
|---|---|---|---|
| (a) | XX–XO method of sex determination | (i) | Turner’s syndrome |
| (b) | XX–XY method of sex determination | (ii) | Female heterogametic |
| (c) | Karyotype – 45 | (iii) | Grasshopper |
| (d) | ZW–ZZ method of sex determination | (iv) | Female homogametic |
Select the correct answer
| Option | (a) | (b) | (c) | (d) |
|---|---|---|---|---|
| 1. | (ii) | (iv) | (i) | (iii) |
| 2. | (i) | (iv) | (ii) | (iii) |
| 3. | (iii) | (iv) | (i) | (ii) |
| 4. | (iv) | (ii) | (i) | (iii) |
Match the following RNA polymerases with their transcribed products:
|
Column-I |
Column-II |
||
|
(a) |
RNA polymerase I |
(i) |
tRNA |
|
(b) |
RNA polymerase II |
(ii) |
rRNA |
|
(c) |
RNA polymerase III |
(iii) |
hnRNA |
Select the correct option from the following:
| Options: | (a) | (b) | (c) |
| 1. | i | iii | ii |
| 2. | i | ii | iii |
| 3. | ii | iii | i |
| 4. | iii | ii | i |
In a marriage between a male with blood group A and a female with blood group B, the progeny had either blood group AB or B. What could be the possible genotype of the parents?
1. IA i (Male) ; IB IB (Female)
2. IA IA (Male) ; IB IB (Female)
3. IA IA (Male) ; IB i (Female)
4. IA i (Male) ; IB i(Female)
A population of a species invades a new area. Which of the following condition will lead to adaptive radiation?
| 1. | Area with a large number of habitats having very low food supply |
| 2. | Area with a single type of vacant habitat |
| 3. | Area with many types of vacant habitats |
| 4. | Area with many habitats occupied by a large number of species |
From the following, identify the correct combination of salient features of the Genetic Code:
| 1. | Universal, Non-ambiguous, Overlapping |
| 2. | Degenerate, Overlapping, Commaless |
| 3. | Universal, Ambiguous, Degenerate |
| 4. | Degenerate, Non-overlapping, Non-ambiguous |
In the process of transcription in Eukaryotes, the RNA polymerase I transcribe -
1. mRNA with additional processing, capping and tailing
2. tRNA, 5 S rRNA and snRNAs
3. rRNAs - 28 S, 18 S and 5.8 S
4. Precursor of mRNA, hnRNA
In which genetic condition, each cell in the affected person, has three sex chromosomes XXY?
1. Thalassemia
2. Klinefelter's Syndrome
3. Phenylketonuria
4. Turner's Syndrome
What initiation and termination factors are involved in transcription in prokaryotes?
1. , respectively
2. , respectively
3. , respectively
4. , respectively
Which of the following statements is correct about the origin and evolution of men?
| 1. | Agriculture came around 50,000 years back. |
| 2. | The Dryopithecus and Ramapithecus primates existing 15 million years ago, walked like men. |
| 3. | Homo habilis probably ate meat. |
| 4. | Neanderthal men lived in Asia between 100000 and 40000 years back. |
The production of gametes by the parents the formation of zygotes, the F1 and F2 plants can be understood using
| 1. | Pie diagram | 2. | A pyramid diagram |
| 3. | Punnett square | 4. | Wenn diagram |
Which of the following statements about methanogens is not correct?
| 1. | They can be used to produce biogas. |
| 2. | They are found in the rumen of cattle and their excreta. |
| 3. | They grow aerobically and breakdown cellulose-rich food. |
| 4. | They produce methane gas. |
Coca alkaloid or cocaine is obtained from:
| 1. | Papaver somniferum | 2. | Atropa belladonna |
| 3. | Erythroxylum coca | 4. | Datura |
Among the following pairs of microbes, which pair has both the microbes that can be used as biofertilizers?
1. Aspergillus and Rhizopus
2. Rhizobium and Rhizopus
3. Cyanobacteria and Rhizobium
4. Aspergillus and Cyanobacteria
Which one of the following cannot be explained on the basis of Mendel’s Law of Dominance
1. Out of one pair of factors one is dominant and the other recessive
2. Alleles do not show any blending and both the characters recover as such in F2 generation
3. Factors occur in pairs
4. The discrete unit controlling a particular character is called a factor
The chromosomes in which centromere is situated close to one end are :
1. Acrocentric
2. Telocentric
3. Sub-metacentric
4. Metacentric
On which organism Beadle and Tatum worked to propose one gene-one enzyme hypothesis
1. Drosophila
2. Escherichia coli
3. Neurospora crassa
4. Nostoc
Amino acid sequence in protein synthesis is determined by sequence of
1. rRNA
2. tRNA
3. mRNA
4. cDNA.
Okazaki fragments during DNA replication
1. Polymerise in 3’ → 5’ direction and form replication fork
2. Prove semiconservative DNA replication
3. Polymerise in 5’ → 3’ direction and explain 3’ → 5’ DNA replication
4. Cause transcription
Read the following four statements (A-D):
(A) In transcription, adenosine pairs with uracil.
(B) Regulation of lac operon by repressor is referred to as positive regulation.
(C) The human genome has approximately 50,000 genes.
(D) Haemophilia is a sex-linked recessive disease.
How many of the above statements are right ?
1. Two
2. Three
3. Four
4. One
Select the correct option:
|
|
Direction of RNA synthesis |
Direction of reading of the template DNA strand |
|
1. |
5' - 3' |
3' - 5' |
|
2. |
3' - 5' |
5' - 3' |
|
3. |
5' - 3' |
5' - 3' |
|
4. |
3' - 5' |
3' - 5' |
Which one of the following is not correctly matched?
1. Aedes aegypti - Yellow fever
2. Anopheles culifacies - Leismaniasis
3. Glossina palpalis - Sleeping sickness
4. Culex pipiens - Filariasis
Which one of the following is the correct statement regarding the particular psychotropic drug specified?
1. Morphine leads to delusions and disturbed emotions
2. Barbiturates cause relaxation and temporary euphoria
3. Hashish causes after thought perceptions and hallucinations
4. Opium stimulates nervous system and causes hallucinations
The active form of Entamaeba-histolytica feeds upon:
1. mucosa and submucosa of colon only
2. food in intestine
3. blood only
4. erythrocytes; mucosa and submucosa of colon
Match each disease with its correct type of vaccine :
|
(a) |
tuberculosis |
(i) |
harmless virus |
|
(b) |
whooping cough |
(ii) |
inactivated toxin |
|
(c) |
diphtheria |
(iii) |
killed bacteria |
|
(d) |
polio |
(iv) |
harmless bacteria |
(a) (b) (c) (d)
1. (iii) (ii) (iv) (i)
2. (iv) (iii) (ii) (i)
3. (i) (ii) (iv) (iii)
4. (ii) (i) (iii) (iv)
Which of the following diseases is caused by a protozoan ?
1. Influenza
2. Babesiosis
3. Blastomycosis
4. Syphilis
Asthma may be attributed to :
| 1. | accumulation of fluid in the lungs |
| 2. | bacterial infection of the lungs |
| 3. | allergic reaction of the mast cells in the lungs |
| 4. | inflammation of the trachea |
For retting of jute the fermenting microbe used is
1. Mesophilic bacteria
2. Helicobacter pylori
3. Butyric acid bacteria
4. Streptococcus lactis
A common biocontrol agent for the control of plant diseases is
1. Trichoderma
2. Baculovirus
3. Bacillus thuringiensis
4. Glomus
An organism used as biofertilizer for raising soyabean crop is
1. Nostoc
2. Azotobacter
3. Azospirillum
4. Rhizobium
Which acts as a promoter for nickel in the hydrogenation of oils?
1. Cu
2. Mo
3. Fe
4. Pt
Given below are three process:
a. Emulsification
b. Adsorption
c. Absorption
The process of froth flotation and chromatography is based on:
1. a & b
2. b & c
3. a & c
4. None of the above
Which pair of elements can form alloy?
1. Zn and Cu
2. Fe and Hg
3. Fe and C
4. C and Pt
In the Hall's process for concentration of bauxite , the ore is fused with:
1. NaHCO3
2. Na2CO3
3. NaF
4. Na3AIF6
The formation of is difficult as compared to because
1. Lone pair of phosphorous is optically inert.
2. Lone pair of phosphorous resides in almost pure p - orbital.
3. Lone pair of phosphorous resides at sp3 orbital.
4. Lone pair of phosphorous resides in almost pure s - orbital.
Incorrectly matched characteristic is:
1. S8: covalent lattice
2. P4: tetrahedron
3. S52-: zigzag
4. SiO2: Covalent lattice
An oxyacid among the following that contains both P-H and
P-P bonds simultaneously is:
1. H4P2O5
2. H4P2O7
3. H4P2O6
4. None of the above
Fe2(CO)9 is diamagnetic.Which of the following reasons is correct?
1. Presence of one CO as bridge group
2. Presence of monodentate ligand
3. Metal-metal(Fe-Fe) bond in molecule
4. Resonance hybridization of CO
10 mg starch protected 5 ml gold sol in 1 ml 2 % NaCl solution. The gold number of starch is:
1. 20
2. 40
3. 80
4. 100
Cr2O72-+XCr3++H2O + oxidized product of X. X in the above reaction cannot be
1. C2O42-
2. Fe2+
3. SO42-
4. S2-
The co-ordination number of Fe in Ferrocene is :
1. 2
2. 5
3. 6
4. 10
Magnetic moment of K3[Fe(CN)5(H2O)] is
1. 0
2. 4.92
3. 3.87
4. 2.83
AgCl on fusion with sodium carbonate, gives:
1.
2.
3.
4.
Correct IUPAC name for is:
1. Hydrogen hexachloroplatinate(IV)
2. Dihydrogen hexachloroplatinate(IV)
3. Hydrogen hexachloroplatinic(IV) acid
4. Hexachloroplatinic(IV) acid
is diamagnetic because:
1. Mn metal is paramagnetic in free state
2. Mn is in +1 oxidation state in this complex
3. NO is present as positive ligand
4. All of the above
Correct sequence of CO bond order in given compounds is:
(P) (Q) CO (R) (S)
1. P>R>S>Q
2. S>P>R>Q
3. Q>S>P>R
4. R>Q>S> P
The EAN of metal atoms in respectively are :
1. 34, 35
2. 34, 36
3. 36, 36
4. 36, 35
The total number of isomers of the complex, complex is :
1. 10
2. 6
3. 4
4. 12
A compound among the following has both fac-mer and optical isomers is:
1. [Co(NH3)5(NO3)]
2. [Co(en)3]3−
3. [Co(gly)3]
4. None of the above
At
1. Decomposes into
2. Decomposes into
3. Sublimes go give liquid
4. Sublimes to give gaseous dimmer
What is produced when orthophosphoric acid reacts with phosphate rock (fluorapatite)?
1.
2.
3.
4.
Heating of Ag with dil. HNO3 give
1. NO
2. NO2
3. N2O
4. N2O3
When excess of SnCl2 is added to HgCl2, the substance formed is
1. Hg2Cl2
2. Sn
3. Hg
4. Cl2
Which of the following elements form stable dinuclear ions
1. Zn
2. Cd
3. Hg
4. Fe
The correct increasing order for the wavelengths of absorption in complexes of Co3+ is:
1. [Co(H2O)6]3+ < [Co(en)3]3+ < [Co(NH3)6]3 +
2. [Co(H2O)6]3+ < [Co(NH3)6]3+ < [Co(en)3]3+
3. [Co(NH3)6]3+ < [Co(en)3]3+ < [Co(H2O)6]3+
4. [Co(en)3]3+ < [Co(NH3)6]3+ < [Co(H2O)6]3+
The correct increasing order of trans-effect of the following species is:
1.
2.
3.
4.
Jahn-Teller effect is not observed in high spin complexes of?
1. d7
2. d8
3. d4
4. d9
1. The magnitude of the charge on the ion alone
2. Size of the ion alone
3. Both magnitude and sign of the charge on the ion.
4. The sign of the charge on the ion alone.
Iron carbonyl, , is classified as:
| 1. | Tetranuclear | 2. | Mononuclear |
| 3. | Trinuclear | 4. | Dinuclear |
The sum of the coordination number and oxidation number of the metal M in the complex [M(en)2(C2O4)]Cl is:
| 1. | Nine (9) | 2. | Six (6) |
| 3. | Seven (7) | 4. | Eight (8) |
In the extraction of copper from its sulphide ore, the metal is finally obtained by the reduction of cuprous oxide with?
1. Iron(II) sulphide
2. Carbon monoxide
3. Copper(I) sulphide
4. Sulphur dioxide
The variation of the boiling point of the hydrogen halides is in the order HF > HI > HBr > HCl.
The higher boiling point of hydrogen fluoride is explained as:
| 1. | The electronegativity of flourine is much higher than other elements in group |
| 2. | There is strong hydrogen bonding between HF molecules |
| 3. | The bond energy of HF molecules is greater than in other hydrogen halides |
| 4. | The effect of nuclear shielding is much reduced in flourine which polarizes the HF molecule |
The strong reducing behaviour of H3PO2 is due to:
1. Presence of one -OH group and two P-H bonds.
2. High electron gain enthalpy of phosphorus.
3. High oxidation state of phosphorus.
4. Presence of two -OH groups and one P-H bond.
"Metals are usually not found as nitrates in their ores". Out of the following two (I and II) reasons which is/are true for the above observation?
| I: | Metal nitrates are highly unstable. |
| II: | Metal nitrates are highly soluble in water. |
Cobalt(III) chloride forms several octahedral complexes with ammonia.
A compound among the following that does not give a test for chloride ions with silver nitrate at 25 °C is:
| 1. | CoCl3. 3NH3 | 2. | CoCl3. 4NH3 |
| 3. | CoCl3. 5NH3 | 4. | CoCl3. 6NH3 |
The correct statement, among the following, is:
| 1. | [Co(CN)6]3- has no unpaired electrons and will be in a low-spin configuration. |
| 2. | [Co(CN)6]3- has four unpaired electrons and will be in a low-spin configuration. |
| 3. | [Co(CN)6]3- has four unpaired electrons and will be In a high-spin configuration. |
| 4. | [Co(CN)6]3- has no unpaired electrons and will be in a high-spin configuration. |
An anticancer agent among the following is:
| 1. | \(\text { mer} - \left[\mathrm{Co}\left(\mathrm{NH}_3\right)_3 \mathrm{Cl}\right] \) | 2. | \(\text { cis -} \left[\mathrm{PtCl}_2\left(\mathrm{NH}_3\right)_2\right] \) |
| 3. | \(\text {cis -} \mathrm{K}_2\left[\mathrm{Pt} \mathrm{Cl}_2 \mathrm{Br}_2\right] \) | 4. | \(\mathrm{NH}_2 \mathrm{CoCl}_4\) |
The reason for the lanthanoid contraction is:
1. negligible screening effect of 'f' orbitals.
2. increasing nuclear charge.
3. decreasing nuclear charge.
4. decreasing screening effect.
The strongest acid among the following compounds is:
| 1. | HClO3 | 2. | HClO4 |
| 3. | H2SO3 | 4. | H2SO4 |
Which of the following statements is false?
| 1. | On passing H2S through acidified K2Cr2O7 solution, a milky colour is observed. |
| 2. | Na2Cr2O7 is preferred over K2Cr2O7 in volumetric analysis. |
| 3. | K2Cr2O7 solution in an acidic medium is orange. |
| 4. | K2Cr2O7 solution becomes yellow in increasing the pH beyond 7 |
| 1. | The greater metallic character of the lanthanoids than that of the corresponding actinoids. |
| 2. | More energy difference between 5f and 6d orbitals than that between 4f and 5d orbitals. |
| 3. | The lesser energy difference between 5f and 6d orbitals than that between 4f and 5d orbitals. |
| 4. | More active nature of the actinoids. |
The electronegativity difference between N and F is greater than that between N and H yet the dipole moment of NH3 (1.5 D) is larger than that of NF3 (0.2 D). This is because:
| 1. | In NH3 as well as in NF3 the atomic dipole and bond dipole are in the same direction |
| 2. | In NH3 the atomic dipole and bond dipole are in the same direction whereas in NF3 these are in opposite directions |
| 3. | In NH3 as well as in NF3 the atomic dipole and bond dipole are in opposite directions |
| 4. | In NH3 the atomic dipole and the bond dipole is in opposite directions whereas in NF3 these are in the same directions |
By the action of concentrated hydrochloric acid on potassium chlorate we get this mixture of gases:
1.
2.
3.
4.
A solution of CuCl in is used to measure the amount of which gas is a sample by simply measuring the change in volume.
1. CO2
2. H2
3. CO
4. All of the above
Magnetic field lines
1. Cannot intersect
2. Are always closed curves
3. Can pass through vacuum
4. All of these
The graph which represents the relation between the total resistance R of a multi-range moving coil voltmeter and its full-scale deflection V is :
1. (i)
2. (ii)
3. (iii)
4. (iv)
For the series LCR circuit shown in the figure, what is the resonance frequency and the amplitude of the current at the resonating frequency ?
1. 2500 rad/s and
2. 2500 rad/s and 5A
3. 2500 rad/s and
4. 25 rad/s and
In a current-carrying long solenoid, the field produced does not depend upon:
| 1. | Number of turns per unit length | 2. | Current flowing |
| 3. | Radius of the solenoid | 4. | All of the above |
In a cyclotron, the angular frequency of a charged particle is independent of :
1. Mass
2. Speed
3. Charge
4. Magnetic field
The basic magnetization curve for a ferromagnetic material is shown in the figure. Then, the value of relative permeability is highest for the point :
1. P
2. Q
3. R
4. S
Two long conductors, separated by a distance d carry current and in the same direction. They exert a force F on each other. Now the current in one of them is increased to two times and its direction is reversed. The distance is also increased to . The new value of the force between them is-
1.
2.
3.
4.
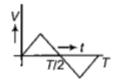
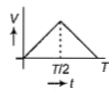
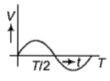
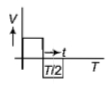
The current (I) in the inductance is varying with time according to the plot shown in figure.
Which one of the following is the correct variation of voltage with time in the coil?
1. 
2. 
3. 
4. 
The electric field part of an electromagnetic wave in a medium is represented by :
1. moving along y-direction with frequency and wavelength 200 m.
2. moving along +x-direction with frequency and wavelength 100 m
3. moving along +x-direction with frequency and wavelength 200 m
4. moving along - x-direction with frequency and wavelength 200 nm
Curie temperature is the temperature above which -
1. ferromagnetic material becomes paramagnetic material
2. paramagnetic material becomes diamagnetic material
3. paramagnetic material becomes ferromagnetic material
4. ferromagnetic material becomes diamagnetic material
A series combination of inductance \((L)\) and resistance \((R)\) is connected to a battery of emf \(E\). The final value of current depends on:
| 1. | \(L\) and \(R\) | 2. | \(E\) and \(R\) |
| 3. | \(E\) and \(L\) | 4. | \(E\), \(L\), and \(R\) |
A circular coil of radius 5 cm has 500 turns of a wire. The approximate value of the coefficient of self induction of the coil will be -
1.
2.
3.
4.
A coil of radius 1 cm and turns 100 is placed in the middle of a long solenoid of radius 5 cm and having 8 turns/cm. The mutual induction in millihenry will be-
1. 0.0316
2. 0.063
3. 0.105
4. Zero
A conducting square loop of side L and resistance R moves in its plane with a uniform velocity v perpendicular to one of its sides.
A magnetic induction B, constant in time and space, pointing perpendicular and into the plane of the loop exists everywhere, see figure. The current induced in the loop is
1. BLv/R clockwise
2. BLv/R anticlockwise
3. 2 BLv/R anticlockwise
4. zero
The magnetic potential energy stored in a certain inductor is \(25~\text{mJ},\) when the current in the inductor is \(60~\text{mA}.\) This inductor is of inductance:
| 1. | \(0.138~\text H\) | 2. | \(138.88~\text H\) |
| 3. | \(1.389~\text H\) | 4. | \(13.89~\text H\) |
If \(\phi_1\) and \(\phi_2\) are the apparent angles of dip observed in two vertical planes at right angles to each other, then the true angle of dip \(\phi\) is given by:
1. \(cos^2{\phi}=cos^2{\phi_1}+cos^2{\phi_2}\)
2. \(sec^2{\phi}=sec^2{\phi_1}+sec^2{\phi_2}\)
3. \(tan^2{\phi}=tan^2{\phi_1}+tan^2{\phi_2}\)
4. \(cot^2{\phi}=cot^2{\phi_1}+cot^2{\phi_2}\)
A bar magnet is hung by a thin cotton thread in a uniform horizontal magnetic field and is in the equilibrium state. The energy required to rotate it by \(60^{\circ}\) is \(W\). Now the torque required to keep the magnet in this new position is:
| 1. | \(\dfrac{W}{\sqrt{3}}\) | 2. | \(\sqrt{3}W\) |
| 3. | \(\dfrac{\sqrt{3}W}{2}\) | 4. | \(\dfrac{2W}{\sqrt{3}}\) |
A uniform magnetic field is restricted within a region of radius \(r\). The magnetic field changes with time at a rate \(\frac{dB}{dt}\). Loop \(1\) of radius \(R>r\) is enclosed within the region \(r\) and loop \(2\) of radius \(R\) is outside the region of the magnetic field as shown in the figure. Then, the emf generated is:
| 1. | zero in loop \(1\) and zero in loop \(2\) |
| 2. | \(-\frac{dB}{dt}\pi r^2\) in loop \(1\) and zero in loop \(2\) |
| 3. | \(-\frac{dB}{dt}\pi R^2\) in loop \(1\) and zero in loop \(2\) |
| 4. | zero in loop \(1\) and not defined in loop \(2\) |
A \(100~\Omega\) resistance and a capacitor of \(100~\Omega\) reactance are connected in series across a \(220~\text{V}\) source. When the capacitor is \(50\%\) charged, the peak value of the displacement current is:
1. \(2.2~\text{A}\)
2. \(11~\text{A}\)
3. \(4.4~\text{A}\)
4. \(11\sqrt{2}~\text{A}\)
In an electromagnetic wave in free space, the root mean square value of the electric field is \(E_{\text{rms}} = 6~\text{V/m}\). The peak value of the magnetic field is:
1. \(2.83\times 10^{-8}~\text{T}\)
2. \(0.70\times 10^{-8}~\text{T}\)
3. \(4.23\times 10^{-8}~\text{T}\)
4. \(1.41\times 10^{-8}~\text{T}\)
An inductor of \(20~\text{mH}\), a capacitor of \(100~\mu \text{F}\), and a resistor of \(50~\Omega\) are connected in series across a source of emf, \(V=10 \sin (314 t)\). What is the power loss in this circuit?
1. \( 0.79 ~\text{W} \)
2. \( 0.43 ~\text{W} \)
3. \( 2.74 ~\text{W} \)
4. \( 1.13 ~\text{W}\)
The current sensitivity of a moving coil galvanometer is \(5~\text{div/mA}\) and its voltage sensitivity (angular deflection per unit voltage applied) is \(20~\text{div/V}.\) The resistance of the galvanometer is:
1. \(40~\Omega\)
2. \(25~\Omega\)
3. \(250~\Omega\)
4. \(500~\Omega\)
Moving perpendicular to field \(B\), a proton and an alpha particle both enter an area of uniform magnetic field \(B\). If the kinetic energy of the proton is \(1~\text{MeV}\) and the radius of the circular orbits for both particles is equal, the energy of the alpha particle will be:
1. \(4~\text{MeV}\)
2. \(0.5~\text{MeV}\)
3. \(1.5~\text{MeV}\)
4. \(1~\text{MeV}\)
A circuit contains an ammeter, a battery of \(30~\text{V},\) and a resistance \(40.8~\Omega\) all connected in series. If the ammeter has a coil of resistance \(480~\Omega\) and a shunt of \(20~\Omega,\) then the reading in the ammeter will be:
1. \(0.5~\text{A}\)
2. \(0.02~\text{A}\)
3. \(2~\text{A}\)
4. \(1~\text{A}\)
| 1. | number of turns in the coil is reduced. |
| 2. | a capacitance of reactance \(X_C = X_L\) is included in the same circuit. |
| 3. | an iron rod is inserted in the coil. |
| 4. | frequency of the AC source is decreased. |
A bar magnet of length \(l\) and magnetic dipole moment \(M\) is bent in the form of an arc as shown in the figure. The new magnetic dipole moment will be:
| 1. | \(\dfrac{3M}{\pi}\) | 2. | \(\dfrac{2M}{l\pi}\) |
| 3. | \(\dfrac{M}{ 2}\) | 4. | \(M\) |
| 1. | can be in equilibrium in one orientation |
| 2. | can be in equilibrium in two orientations, both the equilibrium states are unstable |
| 3. | can be in equilibrium in two orientations, one stable while the other is unstable |
| 4. | experiences a torque whether the field is uniform or non-uniform in all orientations |
The dimensions of where is the permittivity of free space and E is the electric field, are:
1. [ML2T-2]
2. [ML-1T-2]
3. [ML2T-1]
4. [MLT-1]
In the given circuit, the reading of voltmeter V1 and V2 are 300 V each. The reading of the voltmeter V3 and ammeter A are respectively:
1. 150 V, 2.2 A
2. 220 V, 2.2 A
3. 220 V, 2.0 A
4. 100 V, 2.0 A
Electromagnets are made of soft iron because soft iron has:
1. low retentivity and high coercive force
2. high retentivity and high coercive force
3. low retentivity and low coercive force
4. high retentivity and low coercive force
A conducting circular loop is placed in a uniform magnetic field of \(0.04\) T with its plane perpendicular to the magnetic field. The radius of the loop starts shrinking at a rate of \(2\) mm/s. The induced emf in the loop when the radius is \(2\) cm is:
1. \(3.2\pi ~\mu \text{V}\)
2. \(4.8\pi ~\mu\text{V}\)
3. \(0.8\pi ~\mu \text{V}\)
4. \(1.6\pi ~\mu \text{V}\)
| 1. | the rectangular, circular, and elliptical loops. |
| 2. | the circular and the elliptical loops. |
| 3. | only the elliptical loop. |
| 4. | any of the four loops. |
In which of the following devices, the eddy current effect is not used?
1. Electric heater
2. Induction furnace
3. Magnetic braking in train
4. Electromagnet
A parallel plate capacitor of capacitance \(20~\mu\text{F}\) is being charged by a voltage source whose potential is changing at the rate of \(3~\text{V/s}.\) The conduction current through the connecting wires, and the displacement current through the plates of the capacitor would be, respectively:
| 1. | zero, zero | 2. | zero, \(60~\mu\text{A}\) |
| 3. | \(60~\mu\text{A},\) \(60~\mu\text{A}\) | 4. | \(60~\mu\text{A},\) zero |
A step –down transformer transforms a supply line voltage of 2200 volt into 220 volt. The primary coil has 5000 turns. The efficiency and power transmitted by the transformer are 90% and 8 kilowatt respectively. Then the number of turns in the secondary is:
1. 5000
2. 50
3. 500
4. 5
In a certain region, static electric and magnetic field exist. The magnetic field is given by . If a test charge moving with a velocity experiences no force in that region, then the electric field in the region, in SI units is :
1.
2.
3.
4.
The average energy - density of electromagnetic wave given by E = (50 N/C) sin( will be nearly
1.
2.
3.
4.
Two toroids \(1\) and \(2\) have total no. of turns \(200\) and \(100\) respectively with average radii \(40~\text{cm}\) and \(20~\text{cm}\) respectively. If they carry the same current \(i,\) what will be the ratio of the magnetic fields along the two loops?
1. \(1:1\)
2. \(4:1\)
3. \(2:1\)
4. \(1:2\)
| 1. | zero | 2. | \(\dfrac{3\mu_0 i}{32R},~\text{inward}\) |
| 3. | \(\dfrac{3\mu_0 i}{32R},~\text{outward}\) | 4. | \(\dfrac{\mu_0 i}{2R},~\text{inward}\) |
The variation of EMF with time for four types of generators is shown in the figures. Which amongst them can be called AC voltage?
 |
 |
| (a) | (b) |
 |
 |
| (c) | (d) |
| 1. | (a) and (d) |
| 2. | (a), (b), (c), and (d) |
| 3. | (a) and (b) |
| 4. | only (a) |
The relation amongst the three elements of Earth's magnetic field, namely horizontal component H, vertical component V and dip angle is: (=total magnetic field):
1. V=tan, H=
2. V=sin, H=cos
3. V=cos, H=sin
4. V=, H=tan
A circuit when connected to an AC source of \(12~\text{V}\) gives a current of \(0.2~\text{A}\). The same circuit when connected to a DC source of \(12~\text{V}\), gives a current of \(0.4~\text{A}\). The circuit is:
| 1. | series \({LR}\) | 2. | series \({RC}\) |
| 3. | series \({LC}\) | 4. | series \({LCR}\) |
A cycle wheel of radius \(0.5\) m is rotated with a constant angular velocity of \(10\) rad/s in a region of a magnetic field of \(0.1\) T which is perpendicular to the plane of the wheel. The EMF generated between its centre and the rim is:
| 1. | \(0.25\) V | 2. | \(0.125\) V |
| 3. | \(0.5\) V | 4. | zero |
| 1. | \(2.5\times 10^{8}~\text{m/s}\) | 2. | \(3\times 10^{8}~\text{m/s}\) |
| 3. | \(2.08\times 10^{8}~\text{m/s}\) | 4. | \(4.32\times 10^{8}~\text{m/s}\) |