Plants can get along without respiratory organs because of the following except:
| 1. | Each plant part takes care of its own gas exchange needs |
| 2. | Plants do not present great demands for gas exchange. |
| 3. | Gas diffusion in plants occurs easily over long distances in plants. |
| 4. | Each living cell in a plant is located quite close to the surface of the plant. |
The co-factor required for the activity of pyruvate dehydrogenase is:
1. Zinc
2. Magnesium
3. Manganese
4. Copper
In the mitochondrial electron transport system, the term Complex III refers to:
1. NADH dehydrogenase
2. Cytochrome c
3. FADH2
4. Cytochrome bc1 complex
What is the net gain of the number of ATP molecules produced during aerobic respiration of one molecule of glucose?
1. 30
2. 36
3. 38
4. 40
If fatty acids were to be respired, they would first be degraded to:
1. Glucose-6 phosphate
2. Pyruvate
3. Acetyl-CoA
4. Alpha ketoglutarate
The targets of angiotensin II are blood vessels and:
(1) nerves
(2) adrenal cortex
(3) adrenal medulla
(4) kidney nephron
Which section of the nephron follows the ascending limb of the loop of Henle?
1. descending limb of the loop
2. proximal convoluted tubule
3. distal convoluted tubule
4. collecting duct
The concentrations of which plasma electrolyte is not regulated mainly by the kidneys?
1. hydrogen
2. potassium
3. iron
4. Bicarbonate
What masks the active sites for myosin head present on the actin myofilaments?
1. Light meromyosin
2. Heavy meromyosin
3. G – actin
4. Troponin
The muscle can directly use which of the following as a source of energy?
1. Creatine phosphate
2. Glucose
3. ATP
4. Fatty acids
Which layer of human retina stores Vitamin A?
1. Pigment layer
2. Rods
3. Layer of bipolar cells
4. Inner plexiform layer
The biomolecule that cannot act as a chemical messenger [hormone] is:
1. Proteins
2. Steroids
3. Fatty acids
4. Carbohydrate
Which of the following is a storage and release center for neurohormones?
1. Anterior pituitary
2. Posterior pituitary
3. Adrenal medulla
4. Pineal
Which hormones stimulate the production of pancreatic juice and bicarbonate?
1. angiotensin and epinephrine
2. gastrin and insulin
3. cholecystokinin and secretin
4. insulin and glucagon
Which region of the diencephalon controls the ANS and endocrine system and body temperature?
1. epithalamus
2. hypothalamus
3. thalamus
4. infundibulum
The reflex arc ends at:
1. sensory neuron
2. motor neuron
3. effector
4. receptor
The brain stem does not include:
1. medulla
2. pons
3. midbrain
4. cerebral cortex
Adenohypophysis is an endocrine gland that secretes all of the following except:
| 1. | corticotropin | 2. | luteinizing hormone |
| 3. | somatostatin | 4. | somatotropin |
A hormonal disorder that develops when pituitary gland produces too much growth hormone during adulthood is known as:
1. cretinism
2. gigantism
3. Conn’s syndrome
4. Acromegaly
One of the examples of the action of the autonomous nervous system is -
1. Knee-jerk reponse
2. Pupillary reflex
3. Peristalsis of the intestines
4. Swallowing of food
The structure marked as X in the following diagram showing a sagittal section of the human brain would be:
1. Corpora quadrigemina
2. Limbic system
3. Corpus callosum
4. Hippocampal gyrus
A canal called the cerebral aqueduct passes through the:
1. Diencephalon
2. Midbrain
3. III ventricle
4. IV ventricle
All the following are functions of kidneys except:
1. Deactivate vitamin D and stimulation of the activity of osteoclasts.
2. Control of the rate of red blood cell production.
3. help to regulate blood pressure.
4. regulate the composition, volume, and pH of body fluids.
The concentrations of substances in the plasma, in the glomerular filtrate, and in urine differ in what way?
1. Plasma contains the most water, glomerular filtrate contains less water, and urine contains the least.
2. Plasma and glomerular filtrate are virtually identical, but urine contains proportionately more waste products.
3. Plasma differs from glomerular filtrate and urine, which have virtually identical concentrations of substances.
4. All three have the same concentrations of nutrients and waste materials, but differ in the amount of proteinaceous material they contain.
Within the renal tubule, two hormones play a role in determining the final volume and sodium concentration of the urine. The hormone ____ regulates sodium reabsorption, while ______ regulates water reabsorption.
1. aldosterone; antidiuretic hormone (ADH)
2. cortisol; atrial natriuretic peptide
3. renin; angiotensin II
4. antidiuretic hormone (ADH); epinephrine
The mucosa of the bladder is comprised of:
1. smooth muscle
2. squamous epithelium
3. transitional epithelium
4. simple columnar epithelium
How many of the following statement is correct for glycolysis?
(1) It is the only process of respiration in aerobic respiration.
(2) During glycolysis there is partial oxidation of glucose to form one molecule of pyruvic acid.
(3) In glycolysis, there are ten enzyme catalyzed steps.
(4) When PGAL is converted into BPGA, two redox equivalents are removed.
Options :
1. 4
2. 3
3. 2
4. 1
Find out odd statement about electron transport system.
| 1. | It is located in inner mitochondrial membrane and consists of four different complexes. |
| 2. | During this process two mobile carrier that areUQ and Cytc. |
| 3. | Cyt a – a3 contains two copper centeres and acts as terminal acceptor of electron. |
| 4. | O2 acts as a final hydrogen acceptor. |
The form of growth in which new cells are continuously added is known as
1. Diffuse form of growth
2. Open form of growth
3. Continuous form of growth
4. Ring form of growth
Which of the following is not the characteristics of cells present in the meristematic phase
1. Cells densely protoplasmic with a large nucleus
2. Cell wall thin and cellulosic
3. the Increased amount of vacuolation
4. Presence of abundant plasmodesmata connections
The exponential growth can be expressed as w1 = w0ert, where r is?
1. Ability of plant to produce new plant material
2. Efficiency index
3. Growth rate
4. All of these
Find incorrect match
| 1. | Differentiation – specialization |
| 2. | De differentiation – Regain capacity to divide again |
| 3. | Rediffrentiation – Specialisation of dedifferentiated cell |
| 4. | Relative growth – Final size |
A second messenger will not be involved in the action of which of the following hormones on its target cells?
1. Adrenocorticotropin
2. Adrenaline
3. Thyroxin
4. Thyrocalcitonin
The releasing and inhibiting hormones synthesized by hypothalamus are transported from the hypothalamus to the anterior pituitary by way of:
1. the general bloodstream
2. a portal system of blood vessels
3. axons that are present in the pituitary stalk or infundibulum
4. transport carrier proteins present in the CSF
Which of the following correctly describes a negative feedback system?
1. As hormone levels rise, hormone release is promoted.
2. Target organ effects inhibit further hormone release.
3. As hormone levels decrease, hormone release is promoted.
4. As hormone levels decrease, hormone release is inhibited.
A deficiency of both glucocorticoids and mineralocorticoids will cause:
1. Cushing's syndrome
2. Conn’s syndrome
3. Graves' disease
4. Addison's disease
Which of the following hormones will be antagonistic to insulin in its effects on carbohydrate, fat and protein metabolism?
1. Cortisol
2. Glucagon
3. Human Growth Hormone
4. Thyroxin
A potentially life threatening respiratory failure can occur in cases of severe:
1. hyperparathyroidism
2. hyperthyroidism
3. hypothyroidism
4. hypoparathyroidism
An action potential for a given axon is:
1. different in size each time it occurs
2. always the same size
3. larger when the information is to be carried faster
4. smaller when information goes to a gland rather than a muscle
Area of the brain, most involved in maintaining the body's homeostasis, is:
1. medulla oblongata
2. cerebellum
3. pons
4. hypothalamus
Cerebral aqueduct passes through the:
1. Mid brain
2. Diencephalon
3. Hind brain
4. Spinal cord
If production of pyruvic acid by anaerobic metabolism in a muscle is faster than it can be utilized, the surplus is converted to:
1. lactic acid
2. adenosine diphosphate
3. carboxylic acid
4. creatine phosphokinase
Identify the incorrectly matched pair:
| Joint type | Example | |
| 1. | Hinge | Knee |
| 2. | Pivot | Between atlas and axis vertebra |
| 3. | Gliding | Between the metacarpals |
| 4. | Saddle | Between carpal and metacarpal of thumb |
The number of which of the following would normally be equal in human skeleton?
1. Vertebrochondral ribs and digits in both upper limbs
2. Bones in cranium and the facial skeleton
3. Carpals and tarsals
4. Vertebrosternal ribs and digits in one lower limb
The human ribs are termed as ‘bicephalic’ because:
1. They attach to other bones both dorsally and ventrally
2. They have two articulation surfaces on their dorsal end
3. They have two articulation surfaces on their ventral end
4. They articulate with the help of cartilage at both ends
Identify A, B and C in the diagrammatic representation of the mechanism of hormone action.
Select the correct option from the following:
| 1. | A = Steroid Hormone; B = Hormone receptor Complex; C = Protein |
| 2. | A = Protein Hormone; B = Receptor; C = Cyclic AMP |
| 3. | A = Steroid Hormone; B = Receptor; C = Second Messenger |
| 4. | A = Protein Hormone; B = Cyclic AMP; C = Hormone-receptor Complex |
Which of the following statements is not correct?
| 1. | An action potential in an axon does not move backward because the segment behind is in a refractory phase. |
| 2. | Depolarization of hair cells of cochlea results in the opening of the mechanically gated Potassium- ion channels. |
| 3. | Rods are very sensitive and contribute to daylight vision. |
| 4. | In the knee-jerk reflex, stimulus is the stretching of muscle and response is its contraction. |
Match the following joints with the bones involved:
| Column-I | Column-II | ||
| (a) | Gliding joint | (i) | Between carpal and metacarpal of thumb |
| (b) | Hinge joint | (ii) | Between Atlas and Axis |
| (c) | Pivot joint | (iii) | Between the Carpals |
| (d) | Saddle joint | (iv) | Between Humerus and Ulna |
Select the correct option from the following:
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iv) | (i) | (ii) | (iii) |
| 3. | (iv) | (ii) | (iii) | (i) |
| 4. | (i) | (iii) | (ii) | (iv) |
Artificial light, extended work-time and reduced sleep-time disrupt the activity of:
| 1. | Thymus gland | 2. | Pineal gland |
| 3. | Adrenal gland | 4. | Posterior pituitary gland |
Which of the following conditions will stimulate parathyroid gland to release parathyroid hormone?
1. Fall in active Vitamin D levels
2. Fall in blood Ca+2 levels
3. Fall in bone Ca+2 levels
4. Rise in blood Ca+2 levels
Match the following parts of a nephron with their function:
| (a) | Descending limb of Henle's loop | (i) | Reabsorption of salts only |
| (b) | Proximal convoluted tubule | (ii) | Reabsorption of water only |
| (c) | Ascending limb of Henle's loop | (iii) | Conditional reabsorption of sodium ions |
| (d) | Distal convoluted tubule | (iv) | Reabsorption of ions, water, and organic nutrients |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | i | iii | ii | iv |
| 2. | ii | iv | i | iii |
| 3. | i | iv | ii | iii |
| 4. | iv | i | iii | ii |
Match the items in Column - I with those in Column - II:
| Column I | Column II | ||
| (a) | Podocytes | (i) | Crystallised oxalate |
| (b) | Protonephridia | (ii) | Annelids |
| (c) | Nephridia | (iii) | Amphioxus |
| (d) | Renal calculi | (iv) | Filtration slits |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iii) | (ii) | (iv) | (i) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (iv) | (ii) | (iii) | (i) |
Which of the following receptors are specifically responsible for maintenance of balance of body and posture?
1. Basilar membrane and otoliths
2. Hair cells and organ of corti
3. Tectorial membrane and macula
4. Crista ampullaris and macula
Where is the respiratory electron transport system (ETS) located in plants?
1. Mitochondrial matrix
2. Outer mitochondrial membrane
3. Inner mitochondrial membrane
4. Intermembrane space
Removal of shoot tips is a very useful technique to boost the production of tea leaves. This is because:
| 1. | Gibberellins prevent bolting and are inactivated. |
| 2. | Auxins prevent leaf drop at early stages. |
| 3. | Effect of auxins is removed and the growth of lateral buds is enhanced. |
| 4. | Gibberellins delay the senescence of leaves. |
In order to increase the yield of sugarcane crops, which of the following plant growth regulators should be sprayed?
| 1. | Ethylene | 2. | Auxins |
| 3. | Gibberellins | 4. | Cytokinins |
Which one of the following is not a part of a renal pyramid?
1. Convoluted tubules
2. Collecting ducts
3. Loops Henle
4. Peritubular capillaries
Which one of the following statements is correct with respect to salt water balance inside the body of living organisms?
1. When water is not available camels do not produce urine but store urea in tissues.
2. Salmon fish excretes lot of stored salt through gill membrane when in fresh water.
3. Paramecium discharges concentrated salt solution by contractile vacuoles.
4. The body fluids of fresh water animals are generally hypotonic to surrounding water.
Which of the following process of urine formation takes place all along the renal tubule and collecting duct?
1. Ultrafiltration and tubular reabsorption
2. Ultrafiltration and tubular secretion
3. Tubular reabsorption and secretion
4. Anti-current mechanism and reabsorption
In ornithine cycle, which of the following wastes are removed from the blood?
1. CO2 and ammonia
2. Ammonia and urea
3. CO2 and urea
4. Urea and urine
Lack of relaxation between successive stimuli in sustained muscle contraction is known as
1. fatigue 2. tetanus
3. tonus 4. spasm
Sliding filament theory can be best explained as
1. when myofilaments slide pass each other actin filaments shorten while myosin filament do not shorten
2. actin and myosin filaments shorten and slide pass each other
3. actin and myosin filaments do not shorten but rather slide pass each other
4. when myofilament slide pass each other myosin filament shorten while actin filaments do not shorten
The H-Zone in the skeletal muscle fibre is due to
| 1. | the central gap between actin filaments extending through myosin filaments in the A-band |
| 2. | extension of myosin filaments in the central portion of the A-band |
| 3. | the absence of myofibrils in the central portion of A-band |
| 4. | the central gap between myosin filaments in the A-band |
Which of the following pairs of structure is matched with their correct description?
|
|
Structure |
Description |
|
1. |
Cartilage and cornea |
No blood supply but do require oxygen for respiratory need |
|
2. |
Shoulder joint and elbow joint |
Balls and socket type of joint |
|
3. |
Premolars and molars |
20 in all and 3-rooted |
|
4. |
Tibia and fibula |
Both form parts of knee joint |
It is much easier for a small animal to run uphill than for a large animal, because
1. It is easier to carry a small body weight
2. Smaller animals have a higher metabolic rate
3. Small animals have a lower O2 requirement
4. The efficiency of muscles in large animals is less than in the small animals
Good vision depends on adequate intake of carotene rich food.
Select the best option from the following statements.
I. Vitamin-A derivatives are formed from carotene.
II. The photo pigments are embedded in the membrane discs of the inner segment.
III. Retinal is a derivative of vitamin-A.
IV. Retinal is a light absorbing part of all the visual photo pigments.
1. (I) and (II)
2. (I), (III) and (IV)
3. (I) and (III)
4. (II), (III) and (IV)
Destruction of the anterior horn cells of the spinal cord would result in loss of
1. sensory impulses
2. voluntary motor impulses
3. commissural impulses
4. integrating impulses
Injury to vagus nerve in humans is not likely to affect
1. tongue movements
2. gastrointestinal movements
3. pancreatic secretion
4. cardiac movements.
Which part of the human ear plays no role in hearing as such but is otherwise very much required?
1. Eustachian tube
2. Organ of Corti
3. Vestibular apparatus
4. Ear ossicles
Alzheimer disease in humans is associated with the deficiency of
1. dopamine
2. glutamic acid
3. acetylcholine
4. Gamma Amino Butyric Acid (GABA)
A person is wearing spectacles with concave lenses for correcting vision. While not using the glasses, the image of a distant object in his case will be formed.
1. on the blind spot
2. behind the retina
3. in front of the retina
4. on the yellow spot.
One common example of simple reflex is
1. tying your shoe laces while talking to another person and not looking at them
2. watering of mouth at the sight of a favourite food
3. climbing up a stairs in dark without stumbling
4. closing of eyes when strong light is flashed across them.
One of the example of the action of the autonomous nervous system is
1. Knee-jerk response.
2. Pupillary reflex.
3. Peristalsis of the intestines.
4. Swallowing of food.
Which of the following regions of the brain is incorrectly paired with its function?
1. Medulla oblongata-homeostatic control
2. Cerebellum-language comprehension
3. Corpus callosum-communication between the left and right cerebral cortices
4. Cerebrum-calculation and contemplation
Toxic agents present in food which interfere with thyroxin synthesis lead to the development of
1. toxic goitre
2. cretinism
3. simple goitre
4. thyrotoxicosis
Compared to a bull, a bullock is docile because of:
1. higher levels of thyroxin
2. higher levels of cortisone
3. lower levels of blood testosterone
4. lower levels of adrenalin/noradrenalin in its blood
A health disorder that results from the deficiency of thyroxine in adults and characterized by
(i) a low metabolic rate
(ii) increase in body weight
(iii) tendency to retain water in tissues is
1. hypothyroidism
2. simple goitre
3. myxoedema
4. cretinism
The blood calcium level is lowered by the deficiency of:
1. parathormone
2. thyroxine
3. calcitonin
4. Both 1. and 3.
Which hormone causes dilation of blood vessels, increased oxygen consumption, and gluconeogenesis?
1. ACTH
2. Insulin
3. Adrenalin
4. Glucagon
Which one of the following four glands is correctly matched with the accompanying description?
1. Thyroid - hyperactivity in young children causes cretinism
2. Thymus - starts undergoing atrophy after puberty
3. Parathyroid - secrete parathormone which promotes movement of calcium ions from blood into bones during calcification
4. Pancreas - Delta cells of the Islets of Langerhans secrete a hormone which stimulates glycolysis
Parkinson’s disease (characterized by tremors and progessive rigidity of limbs) is caused by degeneration of brain neurons that are involved in moment control and make use of neurotransmitter
1. Norepinephrine
2. Acetylcholine
3. GABA
4. Dopamine
Which one of the following statements is correct?
| 1. | Neurons regulate endocrine activity, but not vice versa. |
| 2. | Endocrine glands regulate neural activity, and nervous system regulates endocrine glands. |
| 3. | Neither hormones control neural activity nor do the neurons control endocrine activity. |
| 4. | Endocrine glands regulate neural activity, but not vice versa |
Which one of the following is an example of negative feedback loop in humans?
1. Secretion of sweat glands and constriction of skin blood vessels when it is too hot.
2. Constriction of skin blood vessels and contraction of skeletal muscles when it is too cold.
3. Secretion of tears after falling of sand particles into the eye.
4. Salivation of mouth at the sight of delicious food.
The amino acid tryptophan is the precursor for the synthesis of
1. Melatonin and serotonin
2. Thyroxine and triiodothyronine
3. Estrogen and progesterone
4. Cortisol and cortisone
Which statement is wrong for Krebs' cycle?
1. There are three points in the cycle where is reduced to NADH +
2. There is one point in the cycle where is reduced to
3. During conversion of succinyl Co-A to succinic acid, a molecule of GTP is synthesised
4. The cycle starts with condensation of acetyl group (acetyl Co-A) with pyruvic acid to yield citric acid
Which one of the following mammalian cells is not capable of metabolising glucose to carbon-dioxide aerobically?
1. White blood cells
2. Unstriated muscle cells
3. Liver cells
4. Red blood cells
The three boxes in this diagram represent the three major bisynthetic pathways in aerobic respiration. Arrow represent net reactants or products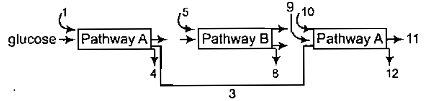
Arrows numbered 4, 8, 12 can all be
1. NADH
2. ATP
3.
4. or
The energy-releasing metabolic process in which substrate is oxidised without an external electron acceptor is called:
| 1. | glycolysis | 2. | fermentation |
| 3. | aerobic respiration | 4. | photorespiration |
During which stage in the complete oxidation of glucose is the greatest number of ATP molecules formed from ADP?
1. Glycolysis
2. Krebs cycle
3. Electron transport
4. Conversion of pyruvic acid to acetyl CoA
Fruit and leaf drop at early stages can be prevented by the application of
1. Cytokinins
2. ethylene
3. auxins
4. gibberellic acid
Cyclopentadiene is much more acidic than cyclopentane, because:
| 1. | Cyclopentadiene has conjugated double bonds. |
| 2. | Cyclopentadiene has both sp2 and sp3 hybridized carbon atoms. |
| 3. | Cyclopentadiene is a strain-free cyclic system. |
| 4. | Cyclopentadienyl anion ion, the conjugate base of cyclopentadiene, is an aromatic species and hence has higher stability. |
 |
The labelled carbon goes with:
Partial oxidation of methane with Ni/Al2O3 catalyst gives:
1. HCHO
2. HCOOH
3. H2O and CO2
4. CO and H2O
A compound among the following that does not undergo Friedel-Craft's reaction easily is:
1. Cumene
2. Xylene
3. Nitrobenzene
4. Toluene
Reaction of alkene and peracid gives oxirane. This reaction is named as-
1. Peroxidation
2. Oxidation
3. epoxydation
4. None
The most probable mechanism for this reaction is-
1. E1
2. E2
3.
4. elimination
What is the correct order of acidity among the following compounds?
| 1. | \(\small\mathrm{CH \equiv CH > CH_3 - C \equiv CH }~ \mathrm{ > CH_2 = CH_2 > CH_3 - CH_3}\) |
| 2. | \(\small\mathrm{CH \equiv CH > CH_2 = CH_2 }~ \mathrm{ > CH_3 - C \equiv CH > CH_3 - CH_3}\) |
| 3. | \(\small\mathrm{CH_3 - CH_3 > CH_2 = CH_2 }~ \mathrm{ > CH_3 - C \equiv CH > CH \equiv CH }\) |
| 4. | \(\small\mathrm{ CH_2 = CH_2 > CH_3 - CH_3 } ~\mathrm{ > CH_3 - C \equiv CH > CH \equiv CH}\) |
The correct statement regarding electrophile is :
| 1. | Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile |
| 2. | Electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile |
| 3. | Electrophiles can be either neutral or positively charged species and can form a bond accepting a pair of electrons from a nucleophile |
| 4. | Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile |
Which among the given molecules can exhibit tautomerism?
1. III only
2. Both I and III
3. Both I and II
4. Both II and III
The compound undergoes the following reactions
C
The product 'C' is-
1. m–Bromotoluene
2. o–Bromotoluene
3. 3–Bromo–2,4,6–trichlorotoluene
4. p–Bromotoluene
The most stable carbocation among the following is:
| 1. |  ` ` |
2. |  |
| 3. |  |
4. |  |
Consider the nitration of benzene using mixed conc. H2SO4 and HNO3.
If a large amount of KHSO4 is added to the mixture, the rate of nitration will be:
| 1. | Slower | 2. | Unchanged |
| 3. | Doubled | 4. | Faster |
Two possible stereo-structures of CH3CHOH.COOH, that are optically active, called:
| 1. | Diastereomers | 2. | Atropisomers |
| 3. | Enantiomers | 4. | Mesomers |
Given compounds are as follows:
| (I) |  |
(II) | 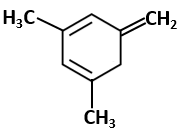 |
| (III) |  |
The enthalpy of hydrogenation of these compounds will be in the order as-
| 1. | I > II > III | 2. | III > II > I |
| 3. | II > III > I | 4. | II > I > III |
Which one of the following is not a common component of Photochemical Smog?
1. Ozone
2. Acrolein
3. Peroxyacetyl nitrate
4. Chlorofluorocarbons
In Kjeldahl’s method for estimation of nitrogen present in the soil sample, ammonia evolved from 0.75g of sample neutralized 10ml of 1M H2SO4. The percentage of nitrogen in the soil is:
| 1. | 37.33 | 2. | 45.85 |
| 3. | 25.75 | 4. | 43.13 |
Which structure corresponds to the IUPAC name 3-Ethyl-2-hydroxy-4-methylhex-3-en-5-ynoic acid?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The correct order of increasing bond length of C-H, C-O, C-C and C=C is:
1.
2.
3.
4.
The Lassaigne’s extract is boiled with conc. while testing for halogens, because it:
1. Helps in the precipitation of
2. Increases the solubility product of
3. Increases the concentration of ions
4. Decomposes and , if formed
The most reactive compound towards electrophilic reagent among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The most stable conformation of n-butane among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Green chemistry means such reactions that:
| 1. | Produce colour during reactions. |
| 2. | Reduce the use and production of hazardous chemicals. |
| 3. | Are related to the depletion of the ozone layer. |
| 4. | Study the reaction in plants. |
CH3— CHCl—CH2—CH3 has a chiral center. Which one of the following represents its R configuration?
1. \(H_{3}C - \underset{H}{\overset{C_{2}H_{5}}{\underset{|}{\overset{|}{C}}}} - {Cl}\)
2. \(H - \underset{Cl}{\overset{C_{2}H_{5}}{\underset{|}{\overset{|}{C}}}} - {CH}_{3}\)
3. \({Cl} - \underset{H}{\overset{C_{2}H_{5}}{\underset{|}{\overset{|}{C}}}} - {CH}_{3}\)
4. \(H - \underset{C_{2}H_{5}}{\overset{{CH}_{3}}{\underset{|}{\overset{|}{C}}}} - {Cl}\)
The IUPAC name of 
1. 3, 4-Dimethylpentanpyl chloride
2. 1-Chloro-1-oxo-2,2-dimethylpentane
3. 2-Ethyl-3-methylbutanoyl chloride
4. 2, 3-Dimethylpentanoyl chloride
The most suitable reagent for the following conversion is-
| 1. | Hg2+/ H+, H2O | 2. | Na/liquid NH3 |
| 3. | H2, Pd/C, quinoline | 4. | Zn/HCl |
The structure of intermediate A in the following reaction is:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
Environmental Planing organization is
1. CSIR
2. CPHERI
3. ICAR
4. NEERI
What is the intensity of sound in normal conversation
1. 10 -20 decibal
2. 30 - 60 decibal
3. 70 - 90 decibal
4. 120 - 150 decibal
Which of the following is absent in polluted water
1. Hydrilla
2. Water hyacinth
3. Larva of stone fly
4. Blue green algae
Soil erosion can be prevented by :
1. Overgrazing
2. Removal of vegetation
3. Afforestation (Plantation)
4. Increasing bird population
The liquefied gas that is used in dry cleaning along with a suitable detergent is-
1. Water gas
2. Petroleum gas
3. NO2
4. CO2
Imines naturally tautomerize to form:
1. Oximes
2. Hydrazones
3. semicarbazones
4. enamines
Why does the equilibrium between keto and enol tautomers lie far to the keto side?
(I) The keto form is more thermodynamically stable.
(II) The enol form is lower enery.
(III) The enol form is more thermodynamically stable.
1. I only
2. III only
3. I and II only
4. II and III only
Which of the following compounds would be most reactive toward a nucleophile?
1. Pentanal
2. 3-pentanone
3. Pentane
4. 2-nonanone
Each of the acyl compounds listed below contains a six-membered ring except:
1. -lactam
2. cyclohexane carboxylic acid
3. -butyrolactone
4. the anhydride formed from intramolecular ring closure of pentanedioic acid.
Which isomer of the following compound is the most stable?
1. 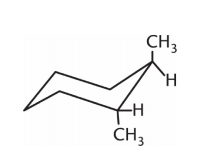
2. 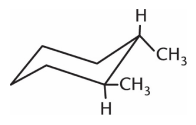
3. 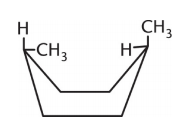
4. They are all equally stable
Consider (E)-2-butene and (Z)-2-butene. This is a pair of what type(s) of isomers?
I. Cis-trans isomers
II. Diastereomers
III. Enantiomers
1. I only
2. II only
3. I and II only
4. I and III only
Rank the following in order of decreasing nucleophilicity in an aprotic solvent:
1.
2.
3.
4.
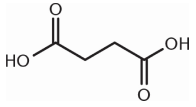
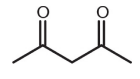
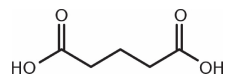
Which of these compounds would be expected to decarboxylate when heated?
1. 
2. 
3. 
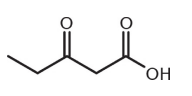
4. 
Carboxylic acids have higher boiling points than their corresponding alcohols primarily because:
1. Molecular weight is increased by the additional carboxyl group
2. the pH of the compound is lower
3. acid salts are soluble in water
4. hydrogen bonding is much stronger than in alcohols
Which of the following carboxylic acids will be the most acidic?
1.
2.
3.
4.
Nucleophilic acyl substitution is favoured by:
I. basic solution
II. acidic solution
III. leaving groups that are strong bases
1. I only
2. II only
3. I and II only
4. I, II, and III
Which of the following amino acids does not have an L-enantiomer?
1. Cysteine
2. Threonine
3. Glutamic acid
4. Glycine
A positive charge on the molecule shown would have greater stability than a positive charge on a straight-chain alkane version of the same molecule. What property most explains this effect?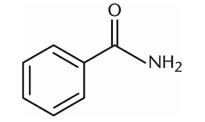
1. Steric hindrane
2. Nitrogen electronegativity
3. Induction
4. Conjugation
The amount of heat energy required to raise the temperature of \(1~\text{g}\) of helium from \(T_1 ~\text{K}\) to\(T_2 ~\text{K}\) is: [Assume volume is constant]
1. \(\dfrac{3}{8}N_Ak_B(T_2-T_1)\)
2. \(\dfrac{3}{2}N_Ak_B(T_2-T_1)\)
3. \(\dfrac{3}{4}N_Ak_B(T_2-T_1)\)
4. \(\dfrac{3}{4}N_Ak_BT_2\)
The displacement of a particle moving in S.H.M. at any instant is given by . The acceleration after time (where T is the time period) -
1.
2.
3.
4.
If the length of second's pendulum is decreased by 2%, how many seconds it will lose per day?
1. 3927 sec
2. 3727 sec
3. 3427 sec
4. 864 sec
A particle with restoring force proportional to displacement and resisting force proportional to velocity is subjected to a force . If the amplitude of the particle is maximum for and the energy of the particle is maximum for , then (where is natural frequency of oscillation of particle)
1. and
2. and
3. and
4. and
The period of oscillation of a simple pendulum of length L suspended from the roof of a vehicle which moves without friction down an inclined plane of inclination , is given by -
1.
2.
3.
4.
The displacement y of a particle executing periodic motion is given by This expression may be considered to be a result of the superposition of ........... independent harmonic motions
1. Two
2. Three
3. Four
4. Five
Three masses, \(700~\text{g},\) \(500~\text{g},\) and \(400~\text{g}\) are suspended vertically from a spring and are in equilibrium. When the \(700~\text{g}\) mass is detached, the remaining system oscillates with a time period of \(3~\text{s}.\) If the \(500~\text{g}\) mass is also removed, what will be the new time period of oscillation?
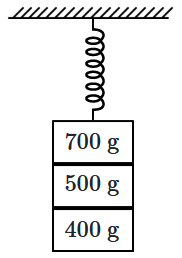
| 1. | \(1~\text{s}\) | 2. | \(2~\text{s}\) |
| 3. | \(3~\text{s}\) | 4. | \(4~\text{s}\) |
A particle of mass m oscillates with simple harmonic motion between points and , the equilibrium position being O. Its potential energy is plotted. It will be as given below in the graph
1. 
2.
3. 
4.
If the equation of the transverse wave is Y = 2sin(kx – 2t), then the maximum particle velocity is :
1. 4 units
2. 2 units
3. 0
4. 6 units
The stationary wave produced on a string is represented by the equation where x and y are in cm and t is in seconds. The distance between consecutive nodes is :
1. 5 cm
2. π cm
3. 3 cm
4. 40 cm
A string is rigidly tied at two ends and its equation of vibration is given by Then minimum length of the string is :
1. 1 m
2.
3. 5 m
4. 2π m
A source of sound placed at the open end of a resonance column sends an acoustic wave of pressure amplitude inside the tube. If the atmospheric pressure is , then the ratio of maximum and minimum pressure at the closed end of the tube will be :
1.
2.
3.
4.
An observer moves towards a stationary source of sound with a speed 1/5th of the speed of sound. The wavelength and frequency of the sound emitted are λ and f respectively. The apparent frequency and wavelength recorded by the observer are respectively :
1.
2.
3.
4.
The equation of displacement of two waves are given as ; . Then what is the ratio of their amplitudes ?
1. 1 : 2
2. 2 : 1
3. 1 : 1
4. None of these
Two identical wires have the same fundamental frequency of 400 Hz when kept under the same tension. If the tension in one wire is increased by 2%, the number of beats produced will be :
1. 4
2. 2
3. 8
4. 1
Two identical pulses in a stretched string, initially \(8\) cm apart, are moving toward each other as shown in the figure. The speed of each pulse is \(2\) cm/s. What will be the total energy of the pulses after \(2~\text{s}\text{?}\)

1. Zero
2. Purely kinetic
3. Purely potential
4. Partly kinetic and partly potential
The displacement-time graphs for two sound waves A and B are shown in the figure, then the ratio of their intensities IA/IB is equal to :
1. 1 : 4
2. 1 : 16
3. 1 : 2
4. 1 : 1
If the ratio of vapour density for hydrogen and oxygen is , then under constant pressure the ratio of their rms velocities will be
1.
2.
3.
4.
A box contains n molecules of a gas. How will the pressure of the gas be effected, if the number of molecules is made 2n?
1. Pressure will decrease
2. Pressure will remain unchanged
3. Pressure will be doubled
4. Pressure will become three times
If the mean free path of atoms is doubled , then the pressure of gas will become
1. P/4
2. P/2
3. P/8
4. P
The kinetic energy of one gram molecules of a gas at standard temperature and pressure:
1.
2.
3.
4.
| 1. | \(L\) | 2. | \(2L\) |
| 3. | \(\dfrac{L}{2}\) | 4. | \(4L\) |
A siren emitting a sound of frequency \(800\) Hz moves away from an observer towards a cliff at a speed of \(15\) ms-1. Then, the frequency of sound that the observer hears in the echo reflected from the cliff is:
(Take, the velocity of sound in air = \(330\) ms-1)
1. \(800\) Hz
2. \(838\) Hz
3. \(885\) Hz
4. \(765\) Hz
A uniform rope, of length \(L\) and mass \(m_1,\) hangs vertically from a rigid support. A block of mass \(m_2\) is attached to the free end of the rope. A transverse pulse of wavelength \(\lambda_1\) is produced at the lower end of the rope. The wavelength of the pulse when it reaches the top of the rope is \(\lambda_2.\) The ratio \(\frac{\lambda_2}{\lambda_1}\) is:
| 1. | \(\sqrt{\dfrac{m_1+m_2}{m_2}}\) | 2. | \(\sqrt{\dfrac{m_2}{m_1}}\) |
| 3. | \(\sqrt{\dfrac{m_1+m_2}{m_1}}\) | 4. | \(\sqrt{\dfrac{m_1}{m_2}}\) |
A source of sound S emitting waves of frequency 100 Hz and an observer O are located at some distance from each other. The source is moving with a speed of 19.4 ms-1 at an angle of with the source-observer line as shown in the figure. The observer is at rest. The apparent frequency observed by the observer (velocity of sound in air 330 ms-1), is:
1. 100 Hz
2. 103 Hz
3. 106 Hz
4. 97 Hz
\(4.0~\text{gm}\) of gas occupies \(22.4~\text{litres}\) at NTP. The specific heat capacity of the gas at a constant volume is \(5.0~\text{JK}^{-1}\text{mol}^{-1}.\) If the speed of sound in the gas at NTP is \(952~\text{ms}^{-1},\) then the molar heat capacity at constant pressure will be:
(\(R=8.31~\text{JK}^{-1}\text{mol}^{-1}\))
| 1. | \(8.0~\text{JK}^{-1}\text{mol}^{-1}\) | 2. | \(7.5~\text{JK}^{-1}\text{mol}^{-1}\) |
| 3. | \(7.0~\text{JK}^{-1}\text{mol}^{-1}\) | 4. | \(8.5~\text{JK}^{-1}\text{mol}^{-1}\) |
| 1. | \( 155~\text{Hz} \) | 2. | \( 205~\text{Hz} \) |
| 3. | \( 10.5~\text{Hz} \) | 4. | \( 105~\text{Hz} \) |
| 1. | \(\dfrac{2}{3}\) | 2. | \(\dfrac{3}{4}\) |
| 3. | \(2\) | 4. | \(\dfrac{1}{2}\) |
A particle is executing a simple harmonic motion. Its maximum acceleration is \(\alpha\) and maximum velocity is \(\beta.\) Then its time period of vibration will be:
1. \(\dfrac {\beta^2}{\alpha^2}\)
2. \(\dfrac {\beta}{\alpha}\)
3. \(\dfrac {\beta^2}{\alpha}\)
4. \(\dfrac {2\pi \beta}{\alpha}\)
When two displacements are represented by \(y_1 = a \text{sin}(\omega t)\) and \(y_2 = b\text{cos}(\omega t)\) are superimposed, then the motion is:
| 1. | not simple harmonic. |
| 2. | simple harmonic with amplitude \(\dfrac{a}{b}\). |
| 3. | simple harmonic with amplitude \(\sqrt{a^2+b^{2}}.\) |
| 4. | simple harmonic with amplitude \(\dfrac{a+b}{2}\). |
If \(n_1\), \(n_2\), and \(n_3\) are the fundamental frequencies of three segments into which a string is divided, then the original fundamental frequency \(n\) of the string is given by:
| 1. | \( \frac{1}{n}=\frac{1}{n_1}+\frac{1}{n_2}+\frac{1}{n_3}\) |
| 2. | \( \frac{1}{\sqrt{n}}=\frac{1}{\sqrt{n_1}}+\frac{1}{\sqrt{n_2}}+\frac{1}{\sqrt{n_3}}\) |
| 3. | \( \sqrt{n}=\sqrt{n_1}+\sqrt{n_2}+\sqrt{n_3}\) |
| 4. | \( n=n_1+n_2+n_3\) |
A point performs simple harmonic oscillation of period \(\mathrm{T}\) and the equation of motion is given by; \(x=a \sin (\omega t+\pi / 6)\). After the elapse of what fraction of the time period, the velocity of the point will be equal to half of its maximum velocity?
1. \( \frac{T}{8} \)
2. \( \frac{T}{6} \)
3. \(\frac{T}{3} \)
4. \( \frac{T}{12}\)
Two points are located at a distance of \(10\) m and \(15\) m from the source of oscillation. The period of oscillation is \(0.05\) s and the velocity of the wave is \(300\) m/s. What is the phase difference between the oscillations of two points?
1. \(\frac{\pi}{3}\)
2. \(\frac{2\pi}{3}\)
3. \(\pi\)
4. \(\frac{\pi}{6}\)
At \(10^{\circ}\text{C}\) the value of the density of a fixed mass of an ideal gas divided by its pressure is \(x.\) At \(110^{\circ}\text{C}\) this ratio is:
| 1. | \(x\) | 2. | \(\dfrac{383}{283}x\) |
| 3. | \(\dfrac{10}{110}x\) | 4. | \(\dfrac{283}{383}x\) |
A mass of \(2.0\) kg is put on a flat pan attached to a vertical spring fixed on the ground as shown in the figure. The mass of the spring and the pan is negligible. When pressed slightly and released, the mass executes a simple harmonic motion. The spring constant is \(200\) N/m. What should be the minimum amplitude of the motion, so that the mass gets detached from the pan?
(Take \(g=10\) m/s2)
| 1. | \(8.0\) cm |
| 2. | \(10.0\) cm |
| 3. | any value less than \(12.0\) cm |
| 4. | \(4.0\) cm |
The phase difference between the instantaneous velocity and acceleration of a particle executing simple harmonic motion is:
1. 0.5
2.
3. 0.707
4. zero
A rectangular block of mass m and area of cross-section A floats in a liquid of density ρ. If it is given a small vertical displacement from equilibrium, it undergoes oscillation with a time period T. Then:
1.
2.
3.
4.
The molar specific heat at a constant pressure of an ideal gas is \(\dfrac{7}{2}R.\) The ratio of specific heat at constant pressure to that at constant volume is:
| 1. | \(\dfrac{7}{5}\) | 2. | \(\dfrac{8}{7}\) |
| 3. | \(\dfrac{5}{7}\) | 4. | \(\dfrac{9}{7}\) |
The amplitude of a simple pendulum, oscillating in air with a small spherical bob, decreases from 10 cm to 8 cm in 40 seconds. Assuming that Stokes law is valid, and the ratio of the coefficient of viscosity of air to that of carbon dioxide is 1.3 , the time in which amplitude of this pendulum will reduce from 10 cm to 5 cm in carbon dioxide will be close to (ln 5 = 1.601, ln 2 = 0.693) :
(1) 231 s
(2) 208 s
(3) 161 s
(4) 142 s
The displacement of a damped harmonic oscillator is given by
. Here t is in seconds. The time taken for its amplitude of vibrations to drop to half of its initial value is close to:
1. 13 s
2. 7 s
3. 27 s
4. 4 s
The distance covered by a particle undergoing SHM in one time period is: (amplitude \(= A\))
1. zero
2. \(A\)
3. \(2 A\)
4. \(4 A\)
| 1. | \(500\) m/s | 2. | \(156\) m/s |
| 3. | \(344\) m/s | 4. | \(172\) m/s |
The value \(\gamma = \frac{C_P}{C_V}\) for hydrogen, helium, and another ideal diatomic gas \(X\) (whose molecules are not rigid but have an additional vibrational mode), are respectively equal to:
| 1. | \(\dfrac{7}{5}, \dfrac{5}{3}, \dfrac{9}{7}\) | 2. | \(\dfrac{5}{3}, \dfrac{7}{5}, \dfrac{9}{7}\) |
| 3. | \(\dfrac{5}{3}, \dfrac{7}{5}, \dfrac{7}{5}\) | 4. | \(\dfrac{7}{5}, \dfrac{5}{3}, \dfrac{7}{5}\) |