Botany - Section A
1. Consider the following statements:
| I: |
Among invertebrates, maximum species richness is seen in insects |
| II: |
Among vertebrates, maximum species richness is seen in mammals |
| III: |
Among plants, algae have a greater diversity than angiosperms. |
1. Only
I is correct
2. Only
II and
III are correct
3. Both
I and
III are correct
4.
I,
II and
III are incorrect
2. The egg apparatus is made up of two ___________ and one _____________ at the ______________ end of the embryo sac.
1. antipodals, egg cell, micropylar
2. synergids, egg cell, chalazal
3. egg cells, synergids, chalazal
4. synergids, egg cells, micropylar
3. An epiphyte is a plant that:
| I: |
grows on other plants for physical support |
| II: |
affects the host negatively |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
4. Filiform apparatus:
| 1. |
is special cellular thickening present in the synergids which play an important role in guiding the pollen tubes into the synergid. |
| 2. |
is special cellular thickening present in the synergids which play an important role in guiding the pollen tubes into the egg cell. |
| 3. |
is special cellular thickening present in the egg cell which play an important role in guiding the pollen tubes into the synergid. |
| 4. |
is special cellular thickening present in the antipodals which play an important role in guiding the pollen tubes into the synergid. |
5. Who proposed the five kingdom classification?
1. Carolus Linnaeus
2. Aristotle
3. Bentham and Hooker
4. R. H. Whittaker
6. A heterogeneous collection of fructose polymers, produced by many types of plants, is:
1. Lectin
2. Ricin
3. Inulin
4. Concanavalin
7.
| A: |
Pollen grains carry the male gamete from microsporangium to micropyle of ovule in angiosperms. |
| B: |
Pollen grain itself a male gamete further divides and form another one on stigma only. |
| 1. |
Both A and B are correct |
2. |
A is correct only |
| 3. |
B is correct only |
4. |
Both A and B are incorrect |
8. Which of the following would be correct regarding the comparison of fermentation and aerobic respiration?
|
Fermentation |
Aerobic respiration |
| I: |
Partial breakdown of glucose |
Complete breakdown of glucose |
| II: |
Net gain of only two molecules of ATP beginning with one molecule of glucose |
Net gain of many more molecules of ATP beginning with one molecule of glucose |
| III: |
NADH is oxidised to NAD+ slowly |
NADH is oxidised to NAD+ vigorously |
| 1. |
Only I and II |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
I, II and III |
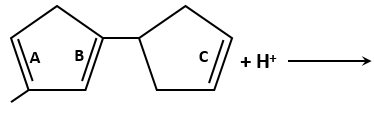
9. As a mechanism of defense against herbivory, Calotropis:
1. Has a large number of thorns
2. Secretes cardiac glycosides
3. Produces a neurotoxin, strychnine
4. Releases a hallucinogen
10. A double-stranded DNA molecule globally has percentage base pair equality: A% = T% and G% = C%. One such DNA molecule is found to have 23 % Adenine. What is the expected % of Uracil in this DNA?
11. A diagram of the transverse section of a monocot leaf is given below with certain parts indicated as letters of the alphabet. Select the option which incorrectly matches the alphabet with the part it represents:

| 1. |
A - Adaxial epidermis |
2. |
B - Phloem |
| 3. |
C - Mesophyll |
4. |
D - Abaxial epidermis |
12. How many of the given statements are true?
| I: |
Amazon rain forest produces 20% of total oxygen in the Earth’s atmosphere |
| II: |
All the biodiversity hotspots put together cover about 10 % of the Earth’s land area |
| III: |
About 23 % of all species of gymnosperms in the world face the threat of extinction |
| IV: |
India’s share of the global species diversity is about 2.4 % |
1. 1
2. 2
3. 3
4. 0
13. Which one of the following is a correct statement?
| 1. |
Pteridophyte gametophyte has a protonemal and leafy stage |
| 2. |
In gymnosperms female gametophyte is free-living |
| 3. |
Antheridiophores and archegoniophores are present in pteridophytes |
| 4. |
Origin of seed habit can be traced in pteridophytes |
14. Identify the incorrect comparison between cellulose and starch:
|
|
Cellulose |
Starch |
| 1. |
Monomer subunit |
Beta Glucose |
Alpha Glucose |
| 2. |
Helical secondary structure |
Yes |
No |
| 3. |
Linkage |
β(1→4)-glycosidic bonds |
α(1→4)-glycosidic bonds |
| 4. |
Type |
Homopolymer |
Homopolymer |
15. Between Telophase I and Prophase II:
| 1. |
The nuclear membrane disappears |
| 2. |
DNA does not replicate |
| 3. |
A tetrad of cells is formed |
| 4. |
There is a long lasting resting stage |
16. Cytochrome c is found:
| 1. |
attached to outer surface of the inner mitochondrial membrane |
| 2. |
attached to inner surface of the inner mitochondrial membrane |
| 3. |
in the inter membrane space |
| 4. |
in the mitochondrial matrix |
17. In which of the following plants, the flowers are epigynous and the position of ovary is described as inferior?
| 1. |
Mustard |
2. |
Rose |
| 3. |
China rose |
4. |
Ray florets of sunflower |
18. Colchicum autumnale:
1. is non-endospermous
2. has dithecous anther
3. has reticulate venation
4. exhibits axile placentation
19. A common biocontrol agent for the control of plant diseases is
1. Trichoderma
2. Baculovirus
3. Bacillus thuringiensis
4. Glomus
20. The phloem parenchyma is absent, and water-containing cavities are present within the vascular bundles in:
| 1. |
Monocot roots |
2. |
Dicot roots |
| 3. |
Monocot stems |
4. |
Dicot stems |
21. Regarding RuBisCO:
| I: |
It is the most abundant protein in the whole of the biosphere. |
| II: |
Mg2+ is its activator |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
22. Which of the following does not take place during diakinesis of Meiosis I?
| 1. |
Disappearance of nucleolus |
| 2. |
Decondensation of chromosomes |
| 3. |
Nuclear envelope breaks down |
| 4. |
Assembly of meiotic spindle |
23. Identify the incorrect statement regarding
Bacillus thuringiensis?
| 1. |
forms protein crystals during a particular phase of their growth which contains a toxic insecticidal protein. |
| 2. |
The Bt toxin protein exists as an inactive protoxin that is converted into an active form by an alkaline pH in the insect's gut. |
| 3. |
Activated toxin binds to the surface of midgut epithelial cells, creates pores, causes cell swelling and lysis and eventually death of the insect. |
| 4. |
Proteins encoded by the genes cryIAc and cryIIAb control the corn borer, that of cryIAb controls cotton bollworms. |
24. T.W. Engelmann:
| 1. |
described a first action spectrum of photosynthesis |
| 2. |
provided evidence for production of glucose when plants grow |
| 3. |
showed that it is only the green parts of the plant that can release oxygen |
| 4. |
first showed that oxygen evolved by the green plant comes from water, not from carbon dioxide |
25. Consider the following two statements:
| I: |
Enzyme catalyzed reactions are affected by the change in the temperature and pH. |
| II: |
Change in pH and temperature affects the tertiary structure of proteins. |
1. Both
I and
II are correct and
II explains
I
2. Both
I and
II are correct but
II does not explain
I
3.
I is correct but
II is incorrect
4.
I is incorrect but
II is correct
26. Match the columns:
|
Column-I |
|
Column-II |
| A. |
Adenine derivative |
(i) |
Ethylene |
| B. |
Terpenes |
(ii) |
Kinetin |
| C. |
Gaseous hormone |
(iii) |
GA |
| D. |
Caroteinoid derivative |
(iv) |
ABA |
| 1. |
A = (i) , B =(ii) , C = (iii) , D= (iv) |
| 2. |
A = (iv) , B =(iii) , C = (i) , D= (ii) |
| 3. |
A = (ii) , B =(iii) , C = (iv) , D= (i) |
| 4. |
A = (ii) , B =(iii) , C = (i) , D= (iv) |
27. Consider the following four statements (A–D), select the correct option stating which ones are true (T) and which ones are false (F)
| (A) |
Organism at each trophic level depends on those at higher trophic levels for their energy demands. |
| (B) |
Each trophic level has a certain mass of living material at a particular time called standing crop. |
| (C) |
The amount of inorganic matter present in an ecosystem at a given time called standing state. |
| (D) |
The number of trophic levels in a grazing food chain is not restricted |
|
A |
B |
C |
D |
| 1. |
T |
T |
F |
F |
| 2. |
F |
T |
F |
T |
| 3. |
T |
F |
T |
F |
| 4. |
F |
T |
T |
F |
28. The Ti Plasmid [ having T-DNA] is considered a natural genetic engineer and can transform:
1. bacterial cells
2. dicot plant cells
3. monocot plant cells
4. animal cells
29. Identify the incorrect comparison between monocots and dicots in general:
|
Character |
Monocots |
Dicots |
| 1. |
Leaves |
Parallel venation |
Reticulate venation |
| 2. |
Roots |
Primary root of short duration, replaced by adventitial roots forming fibrous or fleshy root systems |
Develops from the radicle. Primary root often persists forming strong tap roots and secondary roots |
| 3. |
Plant stem: Vascular bundles |
Ring of primary bundles with cambium, differentiated into cortex and stele |
Numerous scattered bundles in ground parenchyma, cambium mostly absent, no differentiation between cortical and stelar regions |
| 4. |
Flowers |
Parts in threes (trimerous) or multiples of three |
Fours (tetramerous) or fives (pentamerous) |
30. What would be true if you compare the flow of energy and the flow of chemical nutrients in an ecosystem?
| 1. |
Much larger amount of energy flows through the ecosystem than the amount of nutrients |
| 2. |
Energy is recycled, but nutrients are not |
| 3. |
The requirement of nutrients is obligatory for organisms but the requirement of energy is not. |
| 4. |
Nutrients are recycled, but energy is not. |
31. Which of the following statement is true about the difference between
Cycas and
Pinus?
| 1. |
Pinus has coralloid roots associated with N2-fixing cyanobacteria, whereas Cycas has roots with fungal association in the form of mycorrhiza |
| 2. |
Cycas is heterosporous, whereas Pinus is homosporous |
| 3. |
Male cones and female megasporophylls are borne on different trees in Cycas, whereas they are borne on the same tree in Pinus |
| 4. |
Stems of Cycas are branched, whereas Pinus has unbranched stems |
32. The products of non-cyclic photophosphorylation include:
| 1. |
ATP and P700 |
2. |
ATP and NADPH |
| 3. |
ADP and NADP |
4. |
P700 and P680 |
33. Match each item in Column I with one in Column II and select the correct match from the codes given:
|
Group of protozoans |
|
Example |
| A. |
Amoeboid |
P. |
Entamoeba |
| B. |
Flagellated |
Q. |
Paramecium |
| C. |
Ciliated |
R. |
Trypanosoma |
| D. |
Sporozoan |
S. |
Plasmodium |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
S |
P |
R |
| 2. |
S |
Q |
R |
P |
| 3. |
P |
R |
Q |
S |
| 4. |
R |
P |
S |
Q |
34. Match
List-I with
List-II
|
List-I
(Type of cross) |
|
List-II
(Phenotypic ratio) |
| (A) |
Monohybrid cross |
(I) |
1:1 |
| (B) |
Dihybrid cross |
(II) |
1:2:1 |
| (C) |
Incomplete dominance |
(III) |
3:1 |
| (D) |
Test cross |
(IV) |
9:3:3:1 |
Choose the correct answer from the options given below:
| Options: |
(A) |
(B) |
(C) |
(D) |
| 1. |
III |
IV |
II |
I |
| 2. |
II |
IV |
III |
I |
| 3. |
II |
III |
IV |
I |
| 4. |
IV |
III |
I |
II |
35. Following compatible pollination:
| I: |
the pollen germinates on the stigma to produce a pollen tube through one of the germ pores present in the pollen intine. |
| II: |
the contents of the pollen grain move into the pollen tube. |
| III: |
pollen tube grows through the tissues of the stigma and style and reaches the ovary. |
| IV: |
if pollen grains are shed at two-celled condition the generative cell divides and forms the two male gametes during the growth of pollen tube in the stigma. |
1. Only
I,
II and
III are correct
2. Only
I,
III and
IV are correct
3. Only
II,
III and
IV are correct
4.
I,
II,
III and
IV are correct
Botany - Section B
36. Which of the following is the correct description of an anther?
| 1. |
Pollen sac |
| 2. |
The pollen-producing reproductive organ of a flower |
| 3. |
The male gametophyte of angiosperms |
| 4. |
The part of a stamen that contains the pollen |
37. Single-membrane compartment found in plant cells and mainly functioning in the conversion of fat into sugars are:
1. Peroxisomes
2. Glyoxysomes
3. Lysosomes
4. Ribosomes
38. Match each item in
Column-I with the one in
Column-II regarding C4 plants and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Primary acceptor of carbon dioxide |
P. |
RuBP |
| B. |
Cells in which Calvin cycle takes place |
Q. |
PEP |
| C. |
Primary carbon dioxide fixation product |
R. |
Bundle sheath |
| D. |
Example |
S. |
Mesophyll |
|
|
T. |
PGA |
|
|
U. |
OAA |
|
|
V. |
Sorghum |
|
|
W. |
Wheat |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
S |
U |
V |
| 2. |
P |
S |
T |
W |
| 3. |
Q |
R |
U |
V |
| 4. |
P |
R |
T |
W |
39. Who, amongst the following, was the first scientist to discover that plants give off oxygen as a result of splitting water molecules during photosynthesis, not carbon dioxide molecules as thought before?
| 1. |
C. B. Van Niel |
2. |
T. W. Engelmann |
| 3. |
Robert Hill |
4. |
Jan Ingenhousz |
40. Match the terms in Column-I with their correct description in Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Protonema |
P. |
The first stage in development of the gametophyte in mosses. |
| B. |
Prothallus |
Q. |
The photosynthetic gametophyte in ferns. |
| C. |
Seta |
R. |
In bryophytes, the stalk that supports the capsule, if present. |
| D. |
Stipe |
S. |
The stem-like part of the thallus of brown algae such as kelp. |
Codes
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
P |
R |
S |
| 3. |
P |
Q |
S |
R |
| 4. |
Q |
P |
S |
R |
41. Identify the part labelled as 3 in the given diagram:

| 1. |
Micropyle |
2. |
Nucellus |
| 3. |
Funiculus |
4. |
Hilum |
42. What term is used for ATP synthesis coupled to electron transport and proton movement?
1. Chemiosmosis
2. Oxidation
3. Glycolysis
4. Cell respiration
43. India successfully protected itself against biopiracy in a case where in late 1997, the United States Patent and Trademark Office granted a patent to:
1. Co-667
2. Sharbati Sonora
3. Lerma Rojo
4. Basmati
44. Consider the given two statements:
| Statement I: |
In water pollinated plants such as seagrasses, female flowers remain submerged in water. |
| Statement II: |
Pollen grains in such plants are released on to the surface of water. |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
45. In drought-prone areas, sprays of which of the following can be used as anti-transpirant?
| 1. |
ABA |
2. |
GA3 |
| 3. |
IAA |
4. |
Benzyl amino purine
|
46. The detritus food chain:
| I: |
begins with dead organic matter |
| II: |
is the major conduit of energy flow in aquatic ecosystem. |
1. Only I is correct
2. Only II is correct
3. Both I and II are correct
4. Both I and II are incorrect
47. How many ATP molecules are directly synthesized in glycolytic pathway from one glucose molecule?
48. The most thoroughly studied of the known bacteria-plant interactions is the:
| 1. |
Gall formation on certain angiosperms by Agrobacterium |
| 2. |
Nodulation of Sesbania stems by nitrogen-fixing bacteria |
| 3. |
Plant growth stimulation by phosphate– solubilising bacteria |
| 4. |
Cyanobacterial symbiosis with some aquatic ferns |
49. In a plant, genes A, B and C are located on different autosomes. Two plants with genotypes AaBbCc are inter-crossed. The number of phenotypes and genotypes that can occur in their progeny will respectively be:
| 1. |
6 and 8 |
2. |
8 and 9 |
| 3. |
4 and 16 |
4. |
8 and 27 |
50. What acts as the terminal electron acceptor when glucose is completely oxidised inside a cell in the presence of oxygen?
| 1. |
Hydrogen sulphide |
2. |
Water |
| 3. |
Oxygen |
4. |
Pyruvic acid |
Zoology - Section A
51. First heart sound is due to
1. Opening of tri and bi-cuspid valves
2. Closure of tri and bi-cuspid valves
3. Opening of semilunar valves
4. Closure of semilunar valves
52. Which part of the human brain is concerned with the regulation of body temperature?
1. Medulla oblongata
2. Cerebellum
3. Cerebrum
4. Hypothalamus
53. ELISA works on the principle of:
| 1. |
antigen-antibody interaction |
| 2. |
radioactive probe tagging |
| 3. |
culture of microbe in an artificial medium |
| 4. |
histopathology |
54. It is desirable to use restriction enzymes that create sticky ends in recombinant DNA technology procedures because this:
| 1. |
facilitates the action of DNA ligase |
| 2. |
makes selection of recombinant DNA possible |
| 3. |
increases the copy number of rDNA per cell |
| 4. |
will create multiple fragments of the vector DNA |
55. The lac operon can be described as:
| 1. |
an inducible operon with a positive regulation by the repressor protein |
| 2. |
an inducible operon with a negative regulation by the repressor protein |
| 3. |
a repressible operon with a positive regulation by the repressor protein |
| 4. |
a repressible operon with a negative regulation by the repressor protein |
56. Dysentery in humans can generally be caused by:
| I: |
bacterial infections |
| II: |
parasitic infections |
| III: |
viral infections |
1. Only
I and
II
2. Only
II
3. Only
III
4.
I, II and
III
57. Both Molluscs and Echinoderms:
| 1. |
exhibit radial symmetry as adults |
| 2. |
have segmented bodies |
| 3. |
are diploblastic |
| 4. |
are coelomates |
58.
Leydig cells, found adjacent to the seminiferous tubules in the testicle, secrete their hormones when stimulated by:
| 1. |
Androgens |
2. |
LH |
| 3. |
FSH |
4. |
GnRH |
59. The hormone, essential for maintaining pregnancy, secreted in large amounts by the corpus luteum is:
| 1. |
Estrogen |
| 2. |
Progesterone |
| 3. |
LH |
| 4. |
FSH |
60. In frogs:
| I: |
neck and tail are absent. |
| II: |
a pair of nostrils is present. |
| III: |
a membranous tympanum (ear) receives sound signals. |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
61. Which of the following will not be equal in normal physiological conditions?
| 1. |
Partial pressure of oxygen in deoxygenated blood and partial pressure of oxygen in tissues |
| 2. |
Partial pressure of carbon dioxide in alveoli and partial pressure of carbon dioxide in oxygenated blood |
| 3. |
Partial pressure of oxygen in oxygenated blood and partial pressure of oxygen in alveoli |
| 4. |
Partial pressure of carbon dioxide in tissues and partial pressure of carbon dioxide in deoxygenated blood |
62. Oral contraceptive combination pills:
| I: |
Inhibit ovulation |
| II: |
May prevent implantation |
| III: |
Retard passage of sperms through the cervix |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
63. Identify the correct statements about symbiotic relationships:
| I: |
in a parasitic relationship, one organism is harmed and the other is benefited. |
| II: |
coevolution is a feature of many symbiotic relationships. |
| III: |
a relationship that appears to be commensalism may in fact be mutualistic or parasitic. |
| IV: |
the most efficient type of parasite is one that kills its host. |
| 1. |
I, II and III only |
2. |
I, III and IV only |
| 3. |
II, III and IV only |
4. |
I, II, III and IV |
64. Consider the following statements:
| I: |
Glomerulus is a tuft of capillaries formed by the efferent arteriole |
| II: |
Afferent arteriole forms the peritubular capillary network |
| III: |
Vasa recta is absent or highly reduced in juxtamedullary nephrons |
| 1. |
Only III is correct |
| 2. |
I is correct but II and III are incorrect |
| 3. |
I, II and III are incorrect |
| 4. |
I and II are correct |
65. Identify the incorrect statement:
| 1. |
Leukaemias are diagnosed by blood and bone marrow tests. |
| 2. |
CT scan uses X rays. |
| 3. |
MRI uses ionising radiation and strong magnetic fields. |
| 4. |
Antibodies against cancer-specific antigens can be used for the detection of certain cancers. |
66. In a plasmid such as pBR322:
| I: |
Ori controls the copy number of DNA per cell |
| II: |
Multiple cloning sites are recognition sequences of common restriction enzymes |
| III: |
Genes for antibiotic resistance are used as selectable markers |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II, and III are correct |
67. Plasmids are good cloning vectors mainly because:
| 1. |
they are circular |
| 2. |
they replicate autonomously |
| 3. |
they are extra-chromosomal DNA molecules found in many bacterial cells |
| 4. |
they carry genes vital for normal survival and reproduction |
68. Two butterfly species are competing for the same nectar of a flower in a garden. To survive and coexist together, they may avoid competition in the same garden by:
1. feeding at the same time.
2. choosing different foraging patterns.
3. increasing time spent on attacking each other.
4. predating on each other.
69. The thick filaments in the ‘A’ band are also held together in the middle of this band by a thin fibrous membrane called:
| 1. |
I-band |
2. |
Z-line |
| 3. |
M-line |
4. |
Myofibril |
70. All the following statements regarding Medical Termination of Pregnancy are correct except:
| 1. |
It accounts for about 1/5th of the total number of pregnancies conceived in a year |
| 2. |
It was legalized by the Government of India in 1975 |
| 3. |
It is considered safe during the first trimester of pregnancy |
| 4. |
It is legalized induced abortion |
71. At which stage of HIV infection does one usually shows symptoms of AIDS?
| 1. |
When viral DNA is produced by reverse transcriptase. |
| 2. |
When HIV replicates rapidly in helper T-lymphocytes and damages a large number of these. |
| 3. |
Within 15 days of sexual contact with an infected person. |
| 4. |
When the infecting retrovirus enters host cells. |
72. Which of the following will not be an example of passive immunity?
| 1. |
Foetus receiving antibodies from their mother, through placenta. |
| 2. |
Immunity conferred by IgA antibodies present in colostrums. |
| 3. |
Giving antivenom [containing antibodies against venom] after snake bite |
| 4. |
A person developing immunity after natural infection by a pathogen |
73. Match each item in Column I with the one in Column II and select the correct match from the codes given:
|
Column I |
|
Column II |
| A |
Pivot joint |
P |
Between carpal and metacarpal of thumb |
| B. |
Saddle joint |
Q |
Between atlas and axis vertebrae |
| C |
Gliding joint |
R |
Between carpal bones |
| D |
Hinge joint |
S |
Humero-ulnar joint |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
P |
S |
R |
| 3. |
Q |
P |
R |
S |
| 4. |
P |
Q |
S |
R |
74. During post-transcriptional modification of hnRNA in eukaryotes:
| I: |
Capping is done at the 5’ end and tailing at the 3’ end |
| II: |
Exons are removed from the split gene to form the cistron |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
75. The untranslated regions:
| I: |
are present at only the 5’ end of the mature mRNA in eukaryotes |
| II: |
are not required during translation and hence are spliced out of the mature mRNA |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
76. Given below are the pairs of contrasting traits in Pisum sativum as studied by Mendel. Select the incorrectly mentioned option from the table given below:
|
Character |
Dominant |
Recessive |
| 1. |
Flower position |
Terminal |
Axial |
| 2. |
Seeds shape |
Round |
Wrinkled |
| 3. |
Pod colour |
Green |
Yellow |
| 4. |
Pod shape |
Inflated |
Constricted |
77. Which of the following organelles in a eukaryotic cell have circular DNA, 70 S ribosomes and make some of their own proteins?
| 1. |
Golgi apparatus and Vacuole |
| 2. |
Endoplasmic reticulum and Golgi apparatus |
| 3. |
Plastids and mitochondria |
| 4. |
Nucleolus and ribosomes |
78. In the taxonomic categories, which hierarchical arrangement in ascending order is correct in the case of animals?
| 1. |
Kingdom, Order, Phylum, Class, Family, Genus, Species |
| 2. |
Kingdom, Phylum, Class, Order, Family, Genus, Species |
| 3. |
Kingdom, Class, Phylum, Family, Order, Genus, Species |
| 4. |
Kingdom, Order, Class, Phylum, Family, Genus, Species |
79. Which of the following is not true regarding phenylketonuria?
| 1. |
It is inherited as an autosomal recessive condition. |
| 2. |
The affected individual lacks the enzyme phenylalanine hydroxylase. |
| 3. |
The affected individuals lack the capability of converting tyrosine to phenylalanine. |
| 4. |
Clinical features include mental retardation and a reduction in hair and skin pigmentation. |
80. Which diseases given in
Column-I is not correctly matched with sign and symptoms in
Column-II?
|
Column-I |
Column-II |
| 1. |
Filariasis |
Inflammation and swelling in lower limbs and scrotum |
| 2. |
Ringworm |
Internal bleeding, muscular pain, fever, and anaemia |
| 3. |
Amoebiasis |
Abdominal pain, stools with excess mucous, and blood clots |
| 4. |
Pneumonia |
Fever, chills, cough, breathing difficulties |
81. Alec Jeffreys pioneered the technique for:
1. DNA profiling
2. Amplification of DNA
3. Separation of DNA fragments in gel electrophoresis
4. Artificial synthesis of gene
82. The tunica media in veins when compared to the tunic media in arteries:
1. is much thinner
2. is much thicker
3. is equally developed
4. is absent
83. Identify the incorrect statement:
| 1. |
Arrangement of axonemal microtubules in cilia and flagella is described as 9 + 2 |
| 2. |
Arrangement of microtubules in centriole is described as 9 + 0 |
| 3. |
Centrosome is involved in spindle formation in plant cell division |
| 4. |
Centrioles form the basal body of cilia and flagella |
84. Which of the following is not facilitated by LAB?
1. Increasing nutritional quality
2. Increasing vitamin B12 in curd
3. Checking disease causing microbes in intestine
4. Conversion of milk to curd
85. In the human genome:
1. there are 3164.7 billion base pairs
2. largest known gene is HbS
3. more than 20 percent part codes for proteins
4. chromosome 1 has most genes
Zoology - Section B
86. The testes descend into the scrotum during foetal life. Undescended testes are associated with reduced fertility. Which of the following, if true, will explain normal fertility in descended testes?
| 1. |
About 80% of undescended testes descend by the first year of life. |
| 2. |
Most such cases are actually anorchia – an absence of testes. |
| 3. |
The temperature of the testes in the scrotum is at least a few degrees cooler than in the abdomen. |
| 4. |
There is a high rate of anomalies of the epididymis in boys with undescended testes. |
87. The hypothalamus and the pituitary gland mainly control the menstrual cycle. A series of physiological and anatomical processes of puberty culminates in menarche. Which of the following is the most important causal reason for the onset of puberty?
| 1. |
The release of pulses of GnRH by the hypothalamus |
| 2. |
The release of gonadotropins by the anterior pituitary |
| 3. |
Inhibition of GnRH due to increase in circulating gonadotropins |
| 4. |
Secretion of estrogen by the ovaries in response to pituitary hormones |
88. Dictyotene is:
| 1. |
Abnormal meiosis in fungi |
| 2. |
Prolonged resting phase in oogenesis |
| 3. |
Failure of homologues to separate during meiosis |
| 4. |
State of hypermotility in sperms |
89.
The '?' in the given figure shows:

| 1. |
Tight junctions |
2. |
Adhering junctions |
| 3. |
Gap junctions |
4. |
Desmosomes |
90. The spine of the scapula bone is located on its:
| 1. |
lateral border |
2. |
medial border |
| 3. |
ventral surface |
4. |
dorsal surface |
91. The karyotype of a healthy human being will not show:
| 1. |
Metacentric chromosomes |
| 2. |
Submetacentric chromosomes |
| 3. |
Acrocentric chromosomes |
| 4. |
Telocentric chromosomes |
92. In males, the Follicle Stimulating Hormone:
| I: |
stimulates primary spermatocytes to undergo the first division of meiosis, to form secondary spermatocytes. |
| II: |
enhances the production of androgen-binding protein by the Sertoli cells of the testes |
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
93. The decrease in the rate of heartbeat, speed of conduction of action potential and thereby the cardiac output is under the control of
1. Neural signals through the sympathetic nerves
2. Neural signals through the parasympathetic nerves
3. Adrenaline
4. ANS
94. An increase in blood flow to the atria of the heart can cause the release of ANF. ANF:
| I: |
is a potent vasoconstrictor |
| II: |
can cause a decrease in blood pressure |
| III: |
acts as a check on RAAS |
1. Only I and II are correct
2. Only III is correct
3. Only II and III are correct
4. Only I is correct
95. Match each item in Column-I with one in Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Heart |
P. |
Synthesis of angiotensinogen |
| B. |
Lungs |
Q. |
Secretion of atrial natriuretic peptide |
| C. |
Kidneys |
R. |
Primary site for conversion of angiotensin I to angiotensin II |
| D. |
Liver |
S. |
Secretion of renin |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
R |
Q |
P |
S |
| 3. |
Q |
R |
S |
P |
| 4. |
Q |
R |
P |
S |
96. Match the parts of the human brain in Column-I with function in Column-II and select the answer from the codes given:
|
Column-I |
|
Column-II |
| A. |
Medulla oblongata |
a. |
Coordinating center for sensory and motor signaling |
| B. |
Thalamus |
b. |
Regulation of gastric secretions |
| C. |
Hypothalamus |
c. |
Thermoregulation and osmoregulation |
| D. |
Hippocampus |
d. |
Consolidation of short term memory into long term memory |
Codes
|
A |
B |
C |
D |
| 1. |
a |
b |
c |
d |
| 2. |
b |
a |
d |
c |
| 3. |
a |
b |
d |
c |
| 4. |
b |
a |
c |
d |
97. In metamerism, there is serial repetition of unit subdivisions of:
| I. |
Ectoderm products |
| II. |
Mesoderm products |
| III. |
Endoderm products |
| 1. |
Only II |
2. |
Only I and II |
| 3. |
Only II and III |
4. |
I, II and III |
98. A double stranded DNA molecule from a bacteriophage is found to consist of 23% adenine bases. The expected percentage of guanine in this DNA molecule will be:
| 1. |
23 % |
2. |
27 % |
| 3. |
46 % |
4. |
54 % |
99. If the heart rate is 60 per minute, the duration of each cardiac cycle will be:
| 1. |
0.8 seconds |
2. |
0.75 seconds |
| 3. |
1.0 second |
4. |
1.2 second |
100. Match the follwing columns and select the correct option.
| (a) |
Pituitary gland |
(i) |
Grave's disease |
| (b) |
Thyroid gland |
(ii) |
Diabetes mellitus |
| (c) |
Adrenal gland |
(iii) |
Diabetes insipidus |
| (d) |
Pancreas |
(iv) |
Addison's disease |
Options:
|
(a) |
(b) |
(c) |
(d) |
| 1. |
(iii) |
(ii) |
(i) |
(iv) |
| 2. |
(iii) |
(i) |
(iv) |
(ii) |
| 3. |
(ii) |
(i) |
(iv) |
(iii) |
| 4. |
(iv) |
(iii) |
(i) |
(ii) |
Chemistry - Section A
101. In the structure of diborane (B2H6) , the hybridised state of B & the maximum number of atoms which are present in one plane, respectively, are:
| 1. |
sp2 & 4 |
2. |
sp3 & 4 |
| 3. |
sp2 & 6 |
4. |
sp3 & 6 |
102.
| Assertion (A): |
0.1 M solution of glucose has the same decrement in the freezing point as 0.1 M solution of urea. |
| Reason (R): |
Kf for both has the same value. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
103. 
Product (A) in the above mentioned reaction is:
104. The hybridisation of Ni in the complex [Ni(CN)4]2- is:
(Atomic number of Ni = 28)
| 1. |
dsp2 |
2. |
sp3 |
| 3. |
v |
4. |
d2sp3 |
105. Which of the following would have a permanent dipole moment?
| 1. |
BF3 |
2. |
SF4 |
| 3. |
SiF4 |
4. |
XeF4 |
106. When the given compound is hydrated in the presence of
\(H_2SO_4 / HgSO_4, and / H_2O \) , which of the following products is formed?
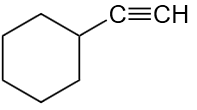
107. In the esterification reaction, H+ ion attacks on:
1. Oxygen atom of alcohol
2. Doubly bonded oxygen atom of carboxylic acid
3. Singly bonded oxygen atom of carboxylic acid
4. Any of the oxygen atoms
108. A liquid that decomposes at its boiling point can be purified by:
| 1. |
Distillation at atmospheric pressure |
| 2. |
Distillation under reduced pressure |
| 3. |
Fractional distillation |
| 4. |
Steam distillation. |
109. At 100 oC the vapour pressure of a solution containing 6.5 g a solute in 100g water is 732mm. If Kb = 0.52 oC m-1, the boiling point of this solution will be:
1. 102C
2. 100C
3. 101C
4. 100.52C
110. The pH of 10-6 M CH3COOH will be:
(Given: ka of CH3COOH = 1.8 \(\times\) 10-5 & log 4.24 =0.63)
1. 5.37
2. 7.0
3. Slightly more than 6
4. 6.95
111. Which is not the product of hydrolysis of XeF6 :
1. XeO3
2. XeO2F2
3. XeOF4
4. XeOF2
112. What is the correct order of ionic mobility of the following ions in an aqueous solution?
| 1. |
K+> Na+> Rb+> Cs+ |
2. |
Cs+> Rb+> K+> Na+ |
| 3. |
Rb+> K+> Cs+> Na+ |
4. |
Na+> K+> Rb+> Cs+ |
113. 
Which \(\pi\) bond is most reactive towards the attack of \(H^+\)?
1. A
2. B
3. C
4. All \(\pi\) bonds have same reactivity
114. Element which has the lowest ionisation potential among the following is:
115. The hybridization of orbitals of N atom in are respectively:
1.
2.
3.
4.
116. Determine the oxidation number of sulfur in Caro's acid (H₂SO₅).
117. For the given reaction:
H2NCOONH4 (s) \( \rightleftharpoons\) 2NH3 (g) + CO2 (g),
Total pressure at equilibrium is found to be 18 atmospheres. The value of Kp for the above equilibrium will be:
| 1. |
72 atm3 |
2. |
144 atm3 |
| 3. |
432 atm3 |
4. |
864 atm3 |
118. Which of the following molecules or ions have one or more lone pairs of valence electrons on their central atom?
| 1. |
\(\mathrm{AlCl_4^-}\) |
2. |
\(\mathrm{CO_2}\) |
| 3. |
\(\mathrm{PCl_4^+}\) |
4. |
\(\mathrm{SO_2}\) |
119. 2.5 litre mixture of CO and CO2 is passed through red hot charcoal in a tube. The new volume becomes 3.5 litre. All measurements are made at same temperature and pressure. Percentage composition of original mixture by volume will be:
1. CO = 50%, CO2 = 50%
2. CO = 60%, CO2 = 40%
3.CO = 40%, CO2 = 60%
4.CO = 20%, CO2 = 80%
120. Which of the following molecules contain bond angles of 180° in their gaseous states?
(Atomic numbers: H = 1; Be = 4; C = 6; Cl = 17)
A. BeCl2
B. C2H2
C. CCl4
| 1. |
A and C only |
2. |
A and B only |
| 3. |
A only |
4. |
B and C only |
121. Case Study:
A buffer solution is defined as a solution whose pH remains practically constant even when small amounts of an acid or a base are added to it.
Types of buffer solutions :
| (i) |
Acidic buffer: It is a solution of a mixture of a weak acid and a salt of this weak acid with a strong base (e.g. CH3COOH + CH3COONa) |
| (ii) |
Basic buffer: It is the solution of a mixture of a weak base and a salt of this weak base with a strong acid (e.g. NH4OH + NH4Cl) |
Henderson's equation is used to determine pH of buffer mixtures of differ types:
for acidic buffer Henderson's equation is
pH= pKa + log \([Salt] \over [Acid]\) (ka = ionisation constant of weak acid )
for basic buffer Henderson's equation is :
POH = Pkb + log \([Salt] \over [Base]\) (kb = ionisation constant of weak base )
When CH3COONa is added to CH3COOH solution, the pH of the solution is:
1. Decreases
2. Increases
3. Remains unchanged
4. First decreases and then increases
122. Insulin is a type of protein. How many amino acids makeup insulin?
123. A saltwater solution contains 23% \(\mathrm{NaCl}\) and 19.5% \(\mathrm{MgCl}_2\) by weight, with both salts fully dissociated (100% dissociation). Calculate the normal boiling point (in ºC) of the saltwater.
(Given : Kb = 0.52 K kg mol–1)
1. 116 oC
2. 121 oC
3. 113 oC
4. 100 oC
124. What is the correct order of electron gain enthalpy from least negative to most negative for the elements C, Ca, Al, F, and O?
1. Al < Ca < O < C < F
2. Al < O < C < Ca < F
3. C < F < O < Al < Ca
4. Ca < Al < C < O < F
125. If 60% of a first-order reaction was completed in 60 min, 50% of the same reaction would be completed in approximately:
(log 4 = 0.60, log 5 = 0.69)
| 1. |
50 min |
2. |
45 min |
| 3. |
60 min |
4. |
40 min |
126. The ionization energy of the hydrogen atom is 13.6 eV and the first ionization energy of the sodium atom is 5.1 eV. The effective nuclear charge experienced by the valence electron of the sodium atom is:
(Round off to one decimal place)
1. 2.8
2. 1.8
3. 1.2
4. 2.2
127. The pair of compounds, from the following pairs, that contains both the compounds with net zero dipole moments is:
1. CH2Cl2 and CHCl3
2. 1,4-dichlorobenzene and 1,3,5-trichlorobenzene
3. Benzene and p-Anisidine
4. Cis-dichloroethene and trans-dichloroethene
128. In aqueous medium, K4[Fe(CN)6] is 40% dissociated. The value of van't Hoff factor (i) for K4[Fe(CN)6] will be:
129. Which of the following compounds has the lowest boiling point?
1.
2.
3.
4.
130. Which of the following compounds has the smallest bond angle?
1. SO2
2. H2O
3. H2S
4. NH3
131. Which of the following pair of elements has same number of unpaired electrons in the ground state?
1. Sulfur (\(S\)) and Chromium (\(Cr\))
2. Nitrogen (\(N\)) and Scandium (\(Sc\))
3. Oxygen (\(O\)) and Manganese(\(Mn\))
4. Arsenic (\(As\)) and Vanadium (\(V\))
132. The correct order of dipole moments for molecules
\(NH_3, H_2S , CH_4 \) and
\(HF\) is :
| 1. |
\(CH_4>H_2S>NH_3>HF \) |
| 2. |
\(H_2S>NH_3>HF>CH_4 \) |
| 3. |
\(NH_3>HF>CH_4>H_2~S \) |
| 4. |
\(HF>NH_3>H_2S>CH_4\) |
133. Identify which of the following compounds readily undergo E2 reactions.
134. Among , the most acidic oxide is:
| 1. |
|
2. |
|
| 3. |
|
4. |
|
135. At equilibrium, which of the following is always zero?
| 1. |
\(\Delta G_{system}\) |
2. |
\(\Delta S_{Total}\) |
| 3. |
\(\Delta S_{system}\) |
4. |
Both (1) and (2) |
Chemistry - Section B
136. An element among the following has the highest first ionization energy is:
137. Calculate the approximate mass of
\(CO_2\) produced when 1 gm of butane
\((C_4H_{10})\) is burned with
an excess of
\(O_2\) to form
\(CO_2\).
| 1. |
1 g |
2. |
2 g |
| 3. |
3 g |
4. |
4 g |
138. The following equilibrium are given
\(N_2+3H_2 \rightleftharpoons 2NH_3~~~........K_1\)
\(N_2+O_2 \rightleftharpoons 2NO~~~.......K_2\)
\(H_2+{1 \over 2}O_2 \rightleftharpoons H_2O~~~........K_3\)
The equilibrium constant of the reaction \(2NH_3+{5 \over 2}O_2 \rightleftharpoons 2NO+3H_2O\), in terms of \(K_1,K_2~\text{and}~K_3~ is :\)
1. \(K_1K_2 \over K_3\)
2. \(K_1K_3^2 \over K_2\)
3. \(K_2K_3^3 \over K_1\)
4. \(K_1K_2K_3\)
139. Correct statement regarding the dichromate ion (Cr2O72–) is:
1. 4 Cr-O bond lengths are equal.
2. 6 Cr-O bond lengths are equal.
3. All Cr-O bond lengths are equal.
4. None of the Cr-O bond lengths are equal.
140. From the given set of 14th group elements, the element having the lowest ionisation potential is:
141. For which of the following processes will the entropy increase?
| 1. |
Reaction of magnesium with oxygen to form magnesium oxide |
| 2. |
Reaction of nitrogen and hydrogen to form ammonia |
| 3. |
Sublimation of dry ice |
| 4. |
Condensation of steam |
142. In Heisenberg's uncertainty principle, if uncertainty in the position
\((\Delta x)\) is equal to the uncertainty in the momentum
\((\Delta p) \) then uncertainty in the velocity
\((\Delta v) \) will be:
| 1. |
\(\dfrac{1}{m}\sqrt{\dfrac{h}{2\pi}}\) |
2. |
\(\dfrac{1}{2m}\sqrt{\dfrac{h}{\pi}}\) |
| 3. |
\(\dfrac{1}{m}\sqrt{\dfrac{h}{\pi}}\) |
4. |
\(\dfrac{1}{2m}\sqrt{\dfrac{h}{2\pi}}\) |
143. 3% (W/V) solution of glucose is isotonic with 1% (W/V) solution of a non-volatile and non-electrolyte substance. The molecular mass of the substance would be:
1. 180 g/mol
2. 360 g/mol
3. 420 g/mol
4. 60 g/mol
144. The molecular formula of a saturated compound is C2H4Br2. This formula permits the existence of:
1. Functional isomers
2. Optical isomers
3. Positional isomers
4. Cis-trans isomers
145. Which is the correct increasing order of the lattice energies for the compounds NaCl, NaF, KCl, and RbCl?
1. KCl < RbCl < NaCl < NaF
2. NaF < NaCl < KCl < RbCl
3. RbCl < KCl < NaCl < NaF
4. NaCl < RbCl < NaF < KCl
146. The following reaction is taking place:
\( 2 \mathrm{NO}+\mathrm{Br}_2 \rightarrow 2 \mathrm{NOBr}\\ \text {Step-01}: \mathrm{NO}+\mathrm{Br}_2 \rightleftharpoons \mathrm{NOBr}_2(fast)\\ \text {Step-02} : \mathrm{NOBr}_2+\mathrm{NO} \rightarrow 2 \mathrm{NOBr} (slow)\)
The order of the reaction is:
1. 1
2. 2
3. 3
4. 4
147. When one mole ethyl methyl ketone is oxidised with acidic K2Cr2O7 then the product formed is:
1. Two moles of CH3COOH
2. One mole of C2H5COOH and one mole of HCOOH
3. One mole of C2H5COOH and one mole of CO2
4. One mole of CH3COOH and 2 mole of HCOOH
148. The IUPAC name of [Pt(NH3)4NO2Cl]SO4 is :
1. Chloronitroplatinum(IV)sulphate
2. Tetraamminechloridonitrito-Nplatinum(IV)sulphate
3. Chloronitrotetraammineplatinum(IV)sulphate
4. Platinum(IV)tetraamminenitrochloro sulphate
149. What mass of copper will be deposited by passing 2 faraday of electricity through a solution of Cu(II) salt?
1. 35.6 g
2. 63.5 g
3. 6.35 g
4. 3.56 g
150. The following data were obtained during the first order thermal decomposition of
\(N_2O_5 (g)\) at constant volume
\(2N_2O_5(g) \rightarrow 2N_2O_4 (g) + O_2 (g) \)
| S. No. |
Time |
Total Pressure (atm) |
| 1. |
0 |
1 |
| 2. |
100 s |
1.25 |
The rate constant of the reaction is:
1. 6.93 x10
1
2. 6.93 x 10
-1
3.
\(6.93 \times 10^{-2}~ \text s \)
4.
\(6.93 \times 10^{-3}~ \text s \)
Physics - Section A
151. \(D_1,D_2\) are ideal diodes and the output
\((V_{AB})\) of the transformer is
\(10\) V (RMS). The current through the resistor is:
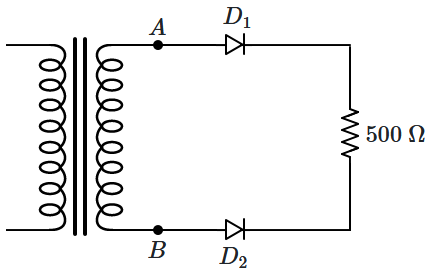
1. zero
2.
\(20\) mA
3.
\(40\) mA
4.
\(20\sqrt2\) mA
152. Photons and electrons of the same wavelength are compared. Which one carries larger momentum?
| 1. |
photon |
| 2. |
electron |
| 3. |
neither, since both have equal momenta |
| 4. |
it could be either, depending on the energy |
153. An ideal monoatomic gas at a temperature of
\(300\) K and a pressure of
\(10\) atm is suddenly allowed to expand into vacuum so that its volume is doubled. No exchange of heat is allowed to take place between the gas and its surroundings during the process. After equilibrium is reached, the final temperature is:
| 1. |
\(300\) K |
2. |
\(\dfrac{300}{2^{5/3}}\) K |
| 3. |
\(\dfrac{300}{2^{2/3}}\) K |
4. |
\(600\) K |
154. A capacitor is constructed by taking metallic circular discs of radius
\(r\) placed face-to-face with a separation of
\(d.\) A dielectric slab is inserted into the space between the plates so that it fills the entire width, but only half the area between the plates. The dielectric constant is
\(K.\) Then, the capacitance is:
| 1. |
\(\dfrac{K\varepsilon_0\pi r^2}{d}\) |
2. |
\(\dfrac{K\varepsilon_0\pi r^2}{2d}\) |
| 3. |
\(\dfrac{K+1}{2}\dfrac{\varepsilon_0\pi r^2}{d}\) |
4. |
None of the above |
155. Plane waves of light of wavelength
\(\lambda\) are incident onto a convex lens, and the beam is brought to a focus. A plane slab of thickness
\(t\) having refractive indices
\(\mu_1,~\mu_2\) in the upper and lower halves is placed parallel to the incoming wavefronts. The phase difference between the wavefronts at the focus, coming from the upper and lower halves of the slab is:

| 1. |
\(\dfrac{2 \pi}{\lambda}\left[\left(\mu_{1}-1\right) t+\left(\mu_{2}-1\right) t\right]\) |
| 2. |
\(\dfrac{2 \pi}{\lambda}\left(\mu_{1}-\mu_{2}\right) t\) |
| 3. |
\(\dfrac{2 \pi}{\lambda}\left(\dfrac{t}{\mu_{1}}-\dfrac{t}{\mu_{2}}\right)\) |
| 4. |
\(\dfrac{2 \pi}{\lambda}\left(\dfrac{t}{\mu_{1}}+\dfrac{t}{\mu_{2}}\right)\) |
156. Given below are two statements:
| Statement I: |
A steady magnetic field can be produced by a steady current. |
| Statement II: |
A steady current can be produced in a circuit by a changing magnetic field. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
157. A light rod
\(AB\) is hinged at
\(A\) so that it is free to rotate about
\(A.\) It is initially horizontal with a small block of mass
\(m\) attached at
\(B,\) and a spring
(constant - \(k\)) holding it vertically up at its mid-point. The time period of vertical oscillations of the system is:

| 1. |
\(2 \pi \sqrt{\dfrac{m}{k}} \) |
2. |
\(\pi \sqrt{\dfrac{m}{k}} \) |
| 3. |
\(4\pi \sqrt{\dfrac{m}{k}}\) |
4. |
\(\dfrac{\pi}{2} \sqrt{\dfrac{m}{k}}\) |
158. The resistance between
\(A,B\) is found to be
\(500~\Omega\) while that between
\(A,C\) is
\(400~\Omega.\) The minimum possible value of
\(R_2\) is:

1.
\(100~\Omega\)
2.
\(200~\Omega\)
3.
\(400~\Omega\)
4.
\(900~\Omega\)
159. Given below are two statements:
| Statement I: |
A nucleus may be left excited after emission of \(\alpha\) or \(\beta\) particles, which then radiates \(\gamma\text-\)ray photons. |
| Statement II: |
Nucleus has discrete energy levels as present in the atom. |
| 1. |
Statement I is false but Statement II is true. |
| 2. |
Both Statement I and Statement II are true. |
| 3. |
Both Statement I and Statement II are false. |
| 4. |
Statement I is true but Statement II is false |
160. A block of wood is immersed in water (assume that the wood is impervious to water), and it floats half immersed. It is placed in an elevator which is accelerating upward. Then,
| 1. |
the block floats, slightly more of it out of the water. |
| 2. |
the block floats, slightly less of it out of the water. |
| 3. |
the block floats, half-immersed. |
| 4. |
the block sinks completely. |
161. A household ac circuit has an applied voltage of
\(220\) V (RMS) and the current flowing through the circuit is
\(2.2\) A (RMS), the phase difference between them being
\(60^\circ.\) Then:
| 1. |
the impedance in the circuit is \(100~\Omega.\) |
| 2. |
the resistance in the circuit is \(200~\Omega.\) |
| 3. |
the power dissipated is \(484\) W. |
| 4. |
all the above are true. |
162. A block of \(1\) kg is released from the top of a smooth curve \(\mathrm{AB},\) and then it encounters a rough surface \(\mathrm{BC},\) coming to rest at \(\mathrm{C}.\) The work done by friction is:
(take \(g=10\) m/s2)

| 1. |
\(25\) J |
2. |
\(50\) J |
| 3. |
\(-25\) J |
4. |
\(-50\) J |
163. A block of mass \(m,\) placed on a rough incline (as shown) – is observed to remain at rest. The coefficient of friction is \(\mu.\) The net force exerted by the incline on the block equals: (in magnitude)

1. \(mg \cos\theta +\mu mg\cos\theta\)
2. \(mg\cos\theta\sqrt{1+\mu^2}\)
3. \(mg\sin\theta\)
4. \(mg\)
164. Two small paramagnetic samples are placed in an (otherwise uniform) strong external magnetic field
\(B.\) If the two samples are placed with a separation that is along the direction of the external field
\(B,\) then, the force exerted by the two samples on each other is:
| 1. |
attractive. |
| 2. |
repulsive. |
| 3. |
zero. |
| 4. |
any of the above depending on the external field \(B\) and the sample separation. |
165. A particle moves around a circle with a unique uniform speed in each revolution. After the first revolution and during the \(2\)nd revolution: its speed doubles; and during the \(3\)rd revolution, its speed becomes \(3\) times the initial speed and so on. The time for the \(1\)st revolution is \(12\) sec. The average time per revolution, for the first four revolutions, is:
1. \(4.8\) s
2. \(9.6\) s
3. \(6.25\) s
4. \(6\) s
166. The radiation emerging from a furnace (blackbody) is found to have a most probable wavelength
\(\lambda_m\) and the gas molecules (air) emerging from it have an RMS speed
\(v.\) As the temperature of the furnace is varied:

| 1. |
\(\lambda_m\propto v \) |
2. |
\(\lambda_m\propto \dfrac1v \) |
| 3. |
\(\lambda_m\propto v^2 \) |
4. |
\(\lambda_m\propto \dfrac1{v^2} \) |
167. A heavy but uniform rope of length \(L\) is suspended from a ceiling. The velocity of a transverse wave travelling on the string as a function of the distance from the lower end is:
1. \(\sqrt{gx}\)
2. \(2\sqrt{gx}\)
3. \(\sqrt{\dfrac{g}{x}} \)
4. \(2\sqrt{\dfrac{g}{x}}\)
168. Which, of the following, is dimensionless?
| 1. |
\(\text{impedance}\times\text{conductance} \) |
2. |
\(\dfrac{\text{emissive power}}{\text{emissivity}}\) |
| 3. |
\(\dfrac{\text{electric field}}{\text{magnetic field}}\) |
4. |
\(\dfrac{\text{inductance}}{\text{capacitance}}\) |
169. In the given circuit,
\(A,\) \(B\) and
\(C\) are the inputs and
\(Y\) is the output. Then
\(Y\) is:
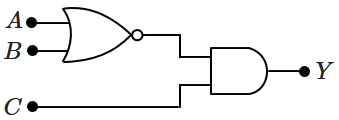
| 1. |
high for all the low inputs. |
| 2. |
high when \(A=1,\) \(B=1,\) \(C=0\). |
| 3. |
low when \(A=0,\) \(B=0,\) \(C=1\). |
| 4. |
low for all low inputs. |
170. A ray of light is incident on a mirror which is lying on the \(x\text-y\) plane. The reflected ray travels along the direction of the vector \(3\hat i-4\hat j+2\hat k. \) The incident ray must be along:
1. \(-3\hat i+4\hat j+2\hat k\)
2. \(3\hat i-4\hat j-2\hat k\)
3. \(-3\hat i-4\hat j+2\hat k\)
4. \(-3\hat i+4\hat j-2\hat k\)
171. At the moment of projection of a projectile (mass: \(m\), initial speed: \(u\), angle of projection: \(\theta\)) the power due to the force of gravity is:
1. \(-mg u\)
2. \(mg u \cos\theta\)
3. \(-mgu \cos^2\theta\)
4. \(-mg u \sin\theta\)
172. A projectile is fired so as to give a maximum horizontal range of
\(1\) km. What would be the maximum height reached by it if it were to be fired vertically upward?
| 1. |
\(2\) km |
2. |
\(1\) km |
| 3. |
\(\dfrac12\) km |
4. |
\(\dfrac14\) km |
173. A thin semi-circular ring of radius
\(r\) has a positive charge
\(q\) distributed uniformly over it. The net potential at the centre
\(O\) is:

| 1. |
\(-\dfrac{q}{2 \pi^{} \varepsilon_{0} r^{}} \) |
2. |
\(-\dfrac{q}{4 \pi^{} \varepsilon_{0} r^{}} \) |
| 3. |
\(\dfrac{q}{4 \pi^{} \varepsilon_{0} r^{}} \) |
4. |
\(0\) |
174. The centre-of-mass of a combination of a hemispherical shell and a cylindrical shell, both having the same height and radii and same mass, lies at a distance \(h\) from the centre of the hemisphere. Then, \(h\) equals:

| 1. |
\(\dfrac{R}{2}\) |
2. |
\(\dfrac{R}{\pi}\) |
| 3. |
\(\dfrac{2R}{\pi}\) |
4. |
zero |
175. The electric field, at the centre of a square with charges placed at its four vertices as shown in the figure, is:
\(\left(k=\dfrac{1}{4\pi\varepsilon_0}\right)\)

| 1. |
zero |
2. |
\(4\dfrac{kq}{a^2}\) |
| 3. |
\(2\dfrac{kq}{a^2}\) |
4. |
\(2\sqrt2\dfrac{kq}{a^2}\) |
176. Two identical capacitors, each of capacitance
\(C\), are connected in series and are charged by means of an ideal battery of emf
\(E\). They are disconnected and reconnected in parallel and connected to the same battery. During this reconnection, the positive terminals of the capacitors are connected to the positive terminal of the battery and their negative terminals are similarly connected together. Let, the work done by the battery during the first connection be
\(W_1\), and during the second be
\(W_2\). Then,
| 1. |
\(W_1=W_2\) |
2. |
\(2W_1 =W_2\) |
| 3. |
\(W_1 = 2W_2\) |
4. |
\(4W_1 = W_2\) |
177. Given below are two statements:
| Assertion (A): |
The pressure within a small air bubble under water is greater than the corresponding pressure outside the bubble. |
| Reason (R): |
This additional pressure within the air bubble is due to the force of the surface tension of water. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
178. A large cylindrical piece of a dense solid elastic metal stands on its end as shown in the figure. The metal is uniform and isotropic. The stress in the material as a function of height is shown correctly by:

179. An equimolar mixture of
helium \(\mathrm{(He)}\) and
hydrogen \(\mathrm{(H_2)}\) gases is kept in a vessel at a temperature of
\(500~\text{K}.\) Then:
| 1. |
helium and hydrogen molecules have the same kinetic energy on average. |
| 2. |
RMS speeds of helium and hydrogen molecules are equal. |
| 3. |
the translational kinetic energy of hydrogen and helium molecules is equal. |
| 4. |
all of the above are true. |
180. The acceleration due to gravity, \(g\), near a spherically symmetric planet's surface decreases with height, \(h\) according to the relation:
\(g(h)= g_s-k\cdot h\), where \(h\ll\) the radius of the planet.
The escape speed from the planet's surface is:
| 1. |
\(\dfrac{g_s}{2\sqrt k}\) |
2. |
\(\dfrac{g_s}{\sqrt k}\) |
| 3. |
\(\dfrac{2g_s}{\sqrt k}\) |
4. |
\(g_s\sqrt{\dfrac{2}{k}} \) |
181. Identical cells are connected to identical square wire loops as shown in the two diagrams, and the magnetic fields are respectively
\(B_1\) and
\(B_2\) at the centres.

Then, we can conclude that:
1.
\(B_1>0, B_2=0 \)
2.
\(B_1> B_2>0 \)
3.
\(B_2> B_1>0 \)
4.
\(B_1=0, B_2=0 \)
182. Photons of wavelength
\(\lambda\) cause the emission of photoelectrons from a metallic surface, the de-Broglie wavelength of the fastest photoelectron being
\(\lambda_d\). A graph of
\(\dfrac{1}{\lambda} \text { vs } \dfrac{1}{\lambda_{d}}\) is:
| 1. |
a straight line passing through the origin. |
| 2. |
a circle. |
| 3. |
an ellipse. |
| 4. |
a parabola. |
183. Given below are two statements:
| Statement I: |
In total internal reflection, the angle of incidence must be greater than a certain minimum angle which depends on the media involved. |
| Statement II: |
Total internal reflection cannot occur when light is traveling from an optically rarer to an optically denser medium. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
184. Light, having a wavelength equal to the first line of the Balmer series, is incident onto a metal of work-function \(2\) eV. The kinetic energy of the ejected electron is:
1. \(1.4\) eV
2. \(0.5\) eV
3. \(0.1\) eV
4. no electrons are ejected
185. An electromagnetic waveform which has an
electric field given by:
\(\vec{E}=E_{0}[\hat{\imath} \cos (\omega t-k z)+\hat{\jmath} \cos (\omega t-k x)]\) and the waveform propagates. The maximum electric field has the magnitude:
| 1. |
\(\dfrac {E_0} { \sqrt 2}\) |
2. |
\(\sqrt 2~ E_0\) |
| 3. |
\(E_o\) |
4. |
\(2E_o\) |
Physics - Section B
186. What is the time period of a simple pendulum with a length of
\(1~\text{m},\) if the acceleration due to gravity at its location is
\(\pi^2~\text{m}\text{s}^{-2} \text{?}\)
| 1. |
\(1~\text{s}\) |
2. |
\(2~\text{s}\) |
| 3. |
\(3~\text{s}\) |
4. |
\(4~\text{s}\) |
187. A long cylindrical solenoid of length
\(L\) and total number of turns
\(N\) produces a magnetic field
\(B_0\) at its centre with a current of
\(1\) A flowing through its coils. If the same current of
\(1\) A is sent through a circular wire of radius
\(R\) then the same field
\(B_0\) is produced at its centre. Then,
\(\dfrac{R}{L}\) equals:
| 1. |
\(N\) |
2. |
\(\dfrac 1N\) |
| 3. |
\(2N\) |
4. |
\(\dfrac{1}{2N}\) |
188. Two blocks of masses \(2m\), \(m\) are placed on a smooth horizontal table and they are in contact on their smooth slanted surfaces. A horizontal force \(F\), equal to \(mg\), is applied to the system from the left, which causes them to accelerate. Let \(N_A\) be the normal reaction from the table on \(A\), and \(N_B\) on \(B\). Then,

| 1. |
\(N_A = 2mg, N_B = mg\) |
| 2. |
\(N_A >2mg, N_B < mg\) |
| 3. |
\(N_A < 2mg, N_B > mg\) |
| 4. |
\(N_A < 2mg, N_B < mg\) |
189. In a Wheatstone's network, \(P = 2~\Omega,Q =2~\Omega\) and \(R = 2~\Omega\) and \(S= 3~\Omega\). The resistance with which \(S\) is to be shunted in order that the bridge may be balanced is:
1. \(1~\Omega\)
2. \(2~\Omega\)
3. \(4~\Omega\)
4. \(6~\Omega\)
190. A uniform cylinder of mass
\(M,\) radius
\(R\) and height
\(3R\) is placed upright on a horizontal surface. A particle of mass
\(m\) is placed on the top of the cylinder at its edge. For what minimum value of
\(m\) will the cylinder topple?

| 1. |
\(m = 3M\) |
| 2. |
\(m= \dfrac {M}{3}\) |
| 3. |
\(m= \dfrac {3M }{2}\) |
| 4. |
No value of \(m\) will cause the cylinder to topple. |
191. A uniform rod of mass \(m\) and length \(L\) is struck at both ends by two particles of masses m, each moving with identical speeds \(u,\) but in opposite directions, perpendicular to its length. The particles stick to the rod after colliding with it. The system rotates with an angular speed:

| 1. |
\(\dfrac{u}{L}\) |
2. |
\(\dfrac{2u}{L}\) |
| 3. |
\(\dfrac{12u}{7L}\) |
4. |
\(\dfrac{6u}{L}\) |
192. A gas is compressed adiabatically. Which of the following statements is not correct?
| 1. |
Internal energy is constant |
| 2. |
Temperature increases |
| 3. |
|Work done| = |change in internal energy| |
| 4. |
Heat is not supplied to the system |
193. In a certain region of space, equipotential surfaces of the electric field are drawn - corresponding to
\(V=10\) volt and \(V=9.9\) volts. There is no field along the
\(z\text-\)direction. At a certain point
\(P,\) on the
\(10-\)volt surface, the distance
\(PQ_1,\) to the
\(9.9\) volt surface is
\(2\) mm when
\(\overrightarrow{P Q}_{1}\) is along the
\(x\text-\)axis. On the other hand, if
\(\overrightarrow{P Q}_{2}\) is taken parallel to the
\(y\text-\)axis, the corresponding distance
\(PQ_2=1\) mm. The electric field at
\(P\) is along:
| 1. |
\(2 \hat{i}+\hat{j}\) |
2. |
\(2 \hat{j}+\hat{i}\) |
| 3. |
\(\dfrac{1}{4} \hat{i}+\hat{j}\) |
4. |
\( \dfrac{1}{4} \hat{j}+\hat{i}\) |
194. The self-inductance of a long solenoid of cross-section
\(A,\) total length
\(L\) and total number of turns
\(N,\) is (approximately):
| 1. |
\(\dfrac{\mu_0A}{L}\cdot N\) |
2. |
\(\dfrac{\mu_0A}{L}\cdot N^2\) |
| 3. |
\(\dfrac{\mu_0L^3}{A}\cdot N\) |
4. |
\(\dfrac{\mu_0L^3}{A}\cdot N^2\) |
195. A parallel beam of light is incident onto a converging lens
\((L_1)\) of focal length
\(20~\text{cm}.\) A second identical lens
\((L_2)\) is to be placed behind
\(L_1,\) coaxially, so that the emerging beam is parallel. The distance between
\(L_1\) and
\(L_2\) should be:

| 1. |
\(10\) cm |
2. |
\(20\) cm |
| 3. |
\(40\) cm |
4. |
zero |
196. Given below are two statements:
| Assertion (A): |
Water flows through a smooth horizontal tube with a narrowing cross-section and its pressure increases. |
| Reason (R): |
Bernoulli's equation for fluids states that \(P+\dfrac{1}{2}\rho v^2+\rho gh= \) constant along a streamline. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
197. Two particles
\(A,B\) move along the periphery of a circle of radius
\(R,\) with the same uniform speed
\(u.\) Particle
\(A\) follows
\(B,\) a quarter of the circumference behind it. The acceleration of
\(A\) relative to
\(B\) is:

| 1. |
zero |
2. |
\(\dfrac{2u^2}{R}\) |
| 3. |
\(\dfrac{u^2}{\sqrt2R}\) |
4. |
\(\dfrac{\sqrt2u^2}{R}\) |
198. Assuming that the diodes
\(D_1\) and
\(D_2\) in the electric circuit are ideal, the value of the current flowing through the
\(1~\Omega\) resistance is:
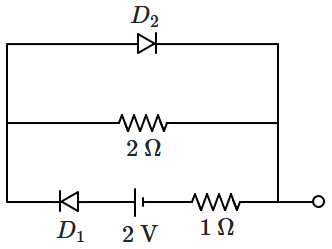
| 1. |
\(\dfrac{2}{3}~\text{A}\) |
2. |
\(2~\text{A}\) |
| 3. |
\(1~\text{A}\) |
4. |
\(\dfrac{1}{2}~\text{A}\) |
199. Two sources of sound vibrating at
\(200~\text{Hz}\) and
\(204~\text{Hz}\) are sounded together. The beat frequency heard is:
| 1. |
\(202~\text{Hz}\) |
2. |
\(404~\text{Hz}\) |
| 3. |
\(4~\text{Hz}\) |
4. |
\(2~\text{Hz}\) |
200. A force \(2x\hat i - 3y^2\hat j\) acts on a particle when it is at the location \(({x, y}).\) This force is:
| 1. |
non-conservative |
| 2. |
conservative and the potential energy is \(({x^2-y^3})\) |
| 3. |
conservative and the potential energy is \(({y^3-x^2})\) |
| 4. |
conservative, but it cannot have a potential energy |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course







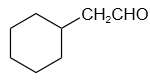
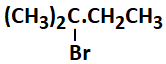
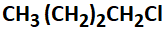
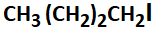
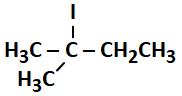
 1. zero
1. zero

















