An organic compound (A) on reduction gives compound (B) which on reaction with chloroform and potassium hydroxide forms (C). The compound (C) on catalytic reduction gives N-methyl aniline. What is the structure of compound (A)?
1. Nitrobenzene
2. Nitromethane
3. Methylamine
4. Aniline
Acetamide and ethyl amine can be distinguished by reacting with:
1. Aq. HCl and heat
2. Aq. NaOH and heat
3. Acidified KMnO4
4. Bromine water
Which of the following compound gives dye test
1. Aniline
2. Methylamine
3. Diphenylamine
4. Ethylamine
Which one of the following on reduction with LiAlH4 yields a secondary amine? [2007]
1. Methyl isocyanide
2. Acetamide
3. Methyl cyanide
4. Nitroethane
Aniline when diazotised in cold and then treated with dimethylaniline, gives a coloured product. Its structure would be :
1. 
2. 
3. 
4. 
What is the end product in the following sequences of operations?
Acetamide
1. CH3NH2
2. C2H5NH2
3. CH3CN
4. CH3COONH4
Name the end product in the following series of reactions,
CH3COOH ABC:
1. CH4
2. CH3OH
3. acetonitrile
4. ammonium acetate
The type of isomerism shown by C6H5CN and C6H5NC is:
1. Position
2. Functional
3. enantiomer
4. Tautomerism
An organic compound A on reduction gives compound B which on reaction with chloroform and potassium hydroxide forms C. The compound C on catalytic reduction gives N-methylaniline. The compound A is [2000]
1. nitrobenzene
2. nitromethane
3. methylamine
4. aniline
An aromatic primary amine with cold nitrous acid leads to the formation of:
1. alcohol
2. nitrite
3. diazonium salt
4. benzene
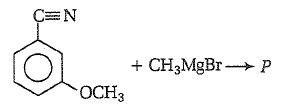

Product P in the above reaction is -
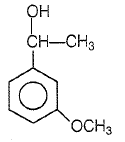
1. 

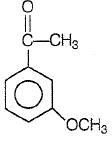
2. 

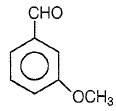
3. 

4. 

In the reaction,
the term Y is
[1999]
1. Acetone
2. Ethanamine
3. Acetaldehyde
4. Dimethyl amine
X and Y in the given reaction are:

1.
2.
3.
4.
Which of the following statements about primary amines is false ?
[2010]
1. Alkyl amines are stronger bases than aryl amines
2. Alkyl amines react with nitrous acid to produce alcohols
3. Aryl amines react with nitrous acid to produce phenols
4. Alkyl amines are stronger bases than ammonia
Among the following, the strongest base is:
1. C6H5NH2
2. p-NO2-C6H4NH2
3. m-NO2-C2H4NH2
4. C2H5CH2NH2
An amine reacts with and the product is soluble in alkali, amine is:
1.
2.
3.
4. all of those
Which of the following reactions does not yield an amine?
1. 
![]()
2. 

3. 
![]()
4. 

The end product in the following sequence of reactions is -
1. Ethyl cyanide
2. Ethylamine
3. Methylamine
4. Acetamide
Identfy X in the sequence,
:
1.
2.
3.
4. none of the above
CH3CH2NH2 contains a basic NH2 group, but CH3CONH2 does not, because;
1. acetamide is amphoteric in character
2. in CH3CH2NH2 the electron pair on N-atom is delocalized by resonance
3. in CH3CH2NH2 there is no resonance, while in acetamide the lone pair of electron on N-atom is delocalized and therefore less available for protonation
4. none of the above
Tertiary nitro compounds cannot show tautomerism because:
1. they are very stable
2. isomerises to give sec. nitro compounds
3. do not have labile H-atom
4. they are highly eactive
Aniline and Ethylamine resemble in:
1. Solubility
2. Action with HNO2
3. Action of Grignard reagent
4. Coupling reaction
A secondary amine is:
1. a compound with two -NH2 groups
2. a compound with 2 carbon atoms and a -NH2 group
3. a compound with a -NH2 group on the carbon atom in number 2 position
4. a compound in which 2 of the hydrogens of NH3 have been replaced by alkyl or aryl groups

The above reaction is an example of....
1. Intermolecular C-N coupling
2. Intramolecular C-N coupling
3. Intermolecular N-N coupling
4. Intramolecular N-N coupling
The hydrochlorides of amines form double salt with:
(1) PtCl4
(2) AuCl3
(3) both (a) and (b)
(4) none of these
An amine gives a solid compound that is insoluble in alkali with benzene sulphonyl chloride.
The amine is -
1.
2.
3.
4.


The compound A and B respectively are:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Reaction of with a mixture of Br2 and KOH gives R-NH2 as the main product. The intermediates involved in this reaction are:
1.
2. RCN
3.
4. RNHBr
In which of the following reactions cyanide will be obtained as a major product?
1.
(A) +

A will be:
(a)
(b)
(c)
(d)
The product formed in the below mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which of the following reactions can be used to prepare acetophenone ?
(a)
(b)
(c)
(d) All of these
Above compounds (P) & (Q) can be differentiated by:
(1) amm. AgNO3
(2) NaOH
(3) FeCl3
(4) Both (a) & (b)
Identify the product (A) in the following reaction series,
CH3CN (X) (Y) (Z) (A):
1. CH3CHO
2. CH3CONH2
3. CH3COOH
4. CH3-CH2-NHOH
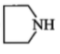
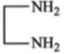
The amine may contain:
(1)
(2)
(3) gp
(4) all of these
Which nitro compound will show tautomerism ?
(1)
(2)
(3)
(4) o-nitrotoluene
The reduction of CH3CN to CH3CH2NH2 is called:
1. Rosenmund's reduction
2. Clemmensen's reduction
3. Mendius reduction
4. Hofmann's reduction
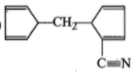
The product [A] formed in the reaction;
1. 
2. 
3. 
4. 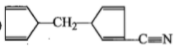
ln the reaction,
The product is:
1. 
2. 
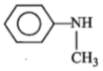
3. 
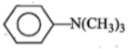
4. 
In the compound given below,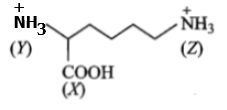
the correct order of acidic nature of the positions (X), (Y) and (Z) is:
(1) Z>X>Y
(2) X>Y>Z
(3) X>Z>Y
(4) Y>X>Z
The product obtained on nitration followed by alkaline hydrolysis of acetylated aniline is:
1. o-Nitroacetanilide
2. o- and p-Nitroaniline
3. m-Nitroaniline
4. Acetanilide
In the chemical reactions,
the compounds 'A' and 'B', respectively, are:
1. Nitrobenzene and chlorobenzene
2. Nitrobenzene and fluorobenzene
3. Phenol and benzene
4. Benzene diazonium chloride and fluorobenzene
Which one of the following nitro-compounds does not react with nitrous acid?
The correct statement regarding the basicity of arylamines is
(1) Arylamines are generally more basic than alkylamines because the nitrogen lone-pair electrons are not delocalized by interaction with the aromatic ring p-electron system.
(2) Arylamines are generally more basic than alkylamines because of aryl group.
(3) Arylamines are generally more basic than alkylamines, because the nitrogen atom in arylamines is sp-hybridized
(4) Arylamines are generally less basic than alkylamines because the nitrogen lone-pair electrons are delocalized by interaction with the aromatic ring p-electron system.
Which of the following is more basic than aniline?
1. Diphenylamine
2. Triphenylamine
3. p-Nitroaniline
4. Benzylamine