The total energy available to the third trophic level organisms if ecosystem is supported by 10 lac J light energy
1. 10 J
2. 100 J
3. 1000 J
4. 10,000 J
Which one of the following trait is only expressed in the presence of identical allele?
1. Yellow seed
2. Inflated pod
3. Green pod
4. Green seed
Blood-cholesterol lowering agent is and produced by
1. Statins, Monascus purpureus
2. Statins, Trichoderma polysporum
3. Cyclosporin A, Streptococcus
4. Streptokinase, Streptococcus
Both chasmogamous and cleistogamous flowers, are produced by
1. Pea and Zostera.
2. Bean and Vallisneria.
3. Maize and bamboo.
4. Oxalis and Commelina.
Common bile duct open into
1. Gall bladder.
2. Duodenum.
3. Hepato-pancreatic duct.
4. Jejunum.
Blood present in afferent arteriole and efferent arteriole will differ in
I. Amount of protein.
II. Number if blood cells.
III. Amount of glucose.
IV. Amount of urea.
1. I, II, III & IV
2. III & IV only
3. IV only
4. III only
Zygomatic and sphenoid bone is an example of
1. Cranial bones.
2. Cranial bone and facial bone respectively.
3. Facial bone and cranial bone respectively.
4. Facial bone.
The vestibular apparatus is composed of:
I. Semi-circular canals
II. Otolith organs
III. Organ of Corti
IV. Crista and macula
1. I, II, III & IV
2. I, II only
3. IV only
4. II only
Low intelligence quotient abnormal skin and deafmutism are related to:
1. Low secretion of growth hormone
2. Hypothyroidism
3. Hyperparathyroidism
4. Hyposecretion of adrenal cortex hormone
Extracellular receptors must be required for the action of:
I. Oxytocin
II. Thyroxine
III. Epinephrine
IV. Glucagon
1. I, II, III & IV
2. I, IV only
3. I, III, IV
4. III & IV only
Mark the correct statement for pBR322 as a plasmid vector
| I: | Contains relaxed origin of replication. |
| II: | Two genes coding for resistance to antibiotics have been introduced. |
| III: | There are single recognition sites for a number of restriction enzymes at various points. |
| IV: | Insertional inactivation is a useful selection method for identifying recombination vectors with insert. |
1. I, II, III & IV
2. I, II only
3. II, III only
4. II only
Level of estrogen and progesterone are minimum at the time of
1. Follicular phase.
2. Ovulation.
3. Secretory phase.
4. Onset of menstrual phase.
Seminal plasma of humans is rich in
1. Fructose and certain enzymes, poor in .
2. Fructose, and certain enzymes.
3. Fructose, but no enzymes.
4. Glucose, certain enzymes but no .
Which one of the following is not the function of placenta?
1. Facilitates removal of carbon dioxide and waste material from embryo.
2. Secrete oxytocin during parturition.
3. Facilitates supply of oxygen and nutrients to embryo.
4. Secretes estrogen.
Diaphragms, cervical caps and vaults prevent conception by
| 1. | Increasing phagocytosis of sperms within uterus. |
| 2. | Suppressing sperm motility. |
| 3. | Inhibiting ovulation and implantation. |
| 4. | Blocking the entry of sperms through the cervix. |
Which plant do not have root modification for support?
1. Banyan.
2. Rhizophora.
3. Sugarcane.
4. Maize.
How many of the following statements are correct for Ribosome?
| 1. | Associated with plasma membrane of cells. |
| 2. | 15-20 nm is size. |
| 3. | Site of protein synthesis. |
| 4. | All of the above. |
When dry weight of tissue is reduced by 10% due to high concentration of a mineral nutrient then
1. It is critical concentration.
2. Mineral nutrient is toxic.
3. Most probably it is macronutrient.
4. It is benificial element.
How many of the following is responsible for seed dormancy?
(1) Abscissic acid.
(2) Phenolic acid.
(3) Para ascorbic acid.
(4) Hard seed coat.
(5) Immature embryo.
(6) GA3
Options
1. 5
2. 6
3. 4
4. 3
In a stamen proximal end of filament is attached to
1. Anther.
2. Thalamus or petals.
3. Sepals.
4. Gynoecium.
Number of ovules in an ovary is more then one in all except
1. Papaya
2. Mango
3. Orchids
4. Water melon
Who established that the behaviour of chromosomes was parallel to the behaviour of gene :-
1. Corners & tschermak.
2. Sutton & Boveri.
3. T.H. Morgan.
4. Klug & cumminis.
In bacteria process of transcription and translation is coupled, because?
1. No seperation of cytosol and nucleus.
2. Ribosome is attached to nuclear envelope.
3. No energy requirement.
4. No cytoplasm.
What is the function of DNA ligase?
1. It synthesises RNA primer.
2. It joins discontinuous strands.
3. It unwinds DNA double strand.
4. It cuts DNA strand.
Match the following
Group Percent facing threat of extinction
A. Birds 1. 12%
B. Mammals 2. 23%
C. Amphbians 3. 32%
D. Gymnosperm 4. 31%
Options
A B C D
1. 4 3 2 1
2. 1 2 3 4
3. 1 3 2 4
4. 1 4 3 2
Identify the incorrectly matched pair:
|
1. |
Ovulation |
LH surge |
|
2. |
Follicular phase of menstrual cycle |
Regeneration of endometrium |
|
3. |
Luteal phase |
Secretion of FSH |
|
4. |
Menstruation |
Withdrawal of progesterone |
Which of the following is not a result of convergent evolution?
1. Potato and sweet potato
2. Flippers of penguins and dolphins
3. Eye of octopus and a mammal
4. Thorn and tendril of Bougainvillea
The process of evolution of different species in a geographical area starting from a point and reaching other areas of habitats is called:
1. Convergent evolution
2. Adaptive radiation
3. Genetic drift
4. Saltation
Identify the incorrectly matched pair:
1. Ramapithecus More man like than ape like
2. Homo habilis First human like being, the hominid
3. Homo erectus Probably did not eat meat
4. Neanderthal Buried their dead
The fetus receives some antibodies from their mother, through the placenta. This is an example of:
1. Naturally acquired passive immunity
2. Artificially acquired passive immunity
3. Naturally acquired active immunity
4. Artificially acquired active immunity
Which of the following drugs has a potent stimulating action of central nervous system?
1. Morphine
2. Cocaine
3. Barbiturate
4. Benzodiazepine
For the multiplication of any alien piece of DNA in an organism:
1. It must be devoid of any introns in it
2. It must be a part of a chromosome with an ori
3. It must have recognition sequence of at least one restriction enzyme
4. It must have selectable markers
In the screening process, the non-recombinant transformed bacteria turn X-gal:
1. Colorless
2. Blue
3. Red
4. Green
Consider the following statements:
I. There is no uncatalyzed metabolic conversion in living systems
II. The living state and metabolism are synonymous
III. All metabolic conversions in living systems are catalyzed by proteins
The number of correct statements is:
1. 0
2. 1
3. 2
4. 3
Identify the given compound:
1. An amino acid
2. A purine base
3. Polysaccharide containing nitrogen
4. Prostaglandin
Dense irregular connective tissue is present in:
1. Skin
2. Tendons
3. Ligaments
4. Bone marrow
Which of the following animals exhibit all these characters – bilateral symmetry, triploblastic, segmented and eucoelomate?
1. Echinoderms
2. Roundworms
3. Molluscs
4. Annelids
What is incorrect regarding cyclostomes?
| 1. | They are ectoparasites on some fishes |
| 2. | They have a circular mouth without jaws |
| 3. | Circulation is of open type |
| 4. | They are marine but migrate to fresh water for spawning |
Identify the incorrectly matched pair:
Animal Characteristic feature
1. Adamsia Cnidocyte
2. Pleurobrachia Comb plates
3. Hook worm Pseudocoelomate
4. Dentalium Water vascular system
Which of the following is not modified mucosal epithelium?
1. Goblet cells
2. Brunner’s glands
3. Crypts of Lieberkuhn
4. Gastric glands
All the following facilitate binding of oxygen to hemoglobin at alveoli except:
1. Low pO2
2. Low pCO2
3. Lesser H+ concentration
4. Lower temperature
During which stage of the cardiac cycle is the first heart sound produced?
| 1. | Atrial diastole | 2. | Joint diastole |
| 3. | Ventricular systole | 4. | Ventricular diastole |
In order to produce concentrated urine, large amounts of water can be reabsorbed from:
1. Ascending limb of loop of Henle
2. Descending limb of loop of Henle
3. Distal convoluted tubule
4. Collecting duct
Identify the incorrectly matched pair:
| Disease | Cause | |
| 1. | Myasthenia gravis | Antibodies against neuromuscular junction |
| 2. | Tetany | Hypocalcemia |
| 3. | Osteoporosis | Hypoparathyroidism |
| 4. | Gout | Accumulation of uric acid crystals |
The resting axonal membrane of a neuron:
1. is not polarized
2. has an excess of anions on the outside
3. is more permeable to potassium ions than to sodium ions
4. cannot be excited by a stimulus
The specific receptor organ in the otolith organ of the internal ear is called:
1. Macula
2. Crista
3. Cupula
4. Organ of Corti
How many of the following are incorrectly matched?
(1) Porphyra - Chlorophyll a and b
(2) Dictyota - Fucoxanthin
(3) Ulothrix - Mannitol
(4) Sargassum - Laminarin
1. 1
2. 3
3. 4
4. 2
Identify the organism that shows the following characteristics.
(a) Zygotic meiosis
(b) Causes red tide of the sea
(c) Mesokaryon organisation
(d) Whirling whips
1. Noctiluca
2. Gonyaulax
3. Melosira
4. Navicula
Select the mismatched pair
Fuits Edible parts
1. Cashew nut Cotyledons
2. Mango Fleshy mesocarp
3. Almond Mesocarp
4. Apple Fleshy thalamus
Read the following statements and identify the incorrect ones.
| (i) | Intrafascicular and interfascicular cambium are the lateral meristems that participate in the secondary growth of dicot stem. |
| (ii) | The heartwood comparises dead elements with highly lignified cell walls. |
| (iii) | Phelloderm has thin walled paranchymatous cells. |
| (iv) | The oldest layer of phloem lies just outside the pericycle. |
| (v) | Phloem paranchyma is absent in most of the monocots. |
1. All except (iv)
2. (i), (ii) and (iii)
3. (iv) only
4. (iii) and (v)
Identify the stages of cell cycle in which the following events occur
(A) Duplication of most of the organelles
(B) Duplication of centrioles
(C) Synthesis of tubulin proteins
(A) (B) (C)
1. G2 phase G1 phase M phase
2. S phase M phase G0 phase
3. M phase Interphase G1 phase
4. G1 phase S phase G2 phase
Leading strand does not require which one of the enzyme during DNA replication?
1. Primase
2. DNA polymerase I
3. DNA polymerase III
4. Ligase
The pure line round seeded pea plant was crossed with wrinkled seeded pea plant. The F1 generation is ____ and it can be explained by _____. (respectively)
1. Wrinkled : Law of segregation
2. Round : law of dominance
3. Round : Condominance
4. Wrinkled : Law of dominance
“Pusa Swarnim” is a/an
1. Aphid resistant variety of Brassica.
2. White rust resistant variety of Brassica.
3. Shoot and fruit borer resistant variety of flat bean.
4. Hill bunt resistant variety of wheat.
“Sexual deceit” is employed by
1. Ophrys to get pollinated.
2. Orchids on mango for shelter.
3. Cuckoo on crow.
4. Abingdon torgoise in Galapagos island.
Humus is
A. Dark coloured amorphous substance.
B. Highly resistant to microbial action.
1. Both A and B are correct.
2. Both A and B are incorrect.
3. A is correct but B is incorrect.
4. A is incorrect but B is correct.
Most important cause for biodiversity loss is
1. Over exploitation.
2. Allien species invasion.
3. Co-extinctions.
4. Habitat loss and fragmentation.
The international treaty to control ozone depletion is
1. Kyoto protocol.
2. Montreal protocol.
3. Bali convention.
4. Earth summit.
Read the following statements and choose the correct pair.
| 1. | Collection of preserved plant and animal specimens – Herbarium. |
| 2. | Solanum, Panthera, Triticum, Felis – Genus. |
| 3. | Reproduction is synonymous to growth – Albugo. |
| 4. | Study of diversity of living organisms along with their evolutionary relationships – Taxonomy. |
Fill in the blanks and choose the correct option
1. Mycelium is coenocytic and aseptate in class ___(i)___
2. ___(ii)___ undergoes rapid multiplication and causes red tide.
3. Sleeping sickness is caused by ___(iii)___ protozoan.
4. The protein coat of virus consist of small subunits called ___(iv)___
W.M. Stanley
1. Discovered TMV.
2. Coined the term virus.
3. Demonstrated that extract of infected plant can cause tobacco mosaic disease in healthy plants.
4. Crystallised virus and showed that crystals are largely made up of proteins.
Read the following and choose the incorrect statement
1. Majority of red algae are marine with greater concentrations found in the warmer areas.
2. Thallus of Marchantia is dorsiventral and closely appressed to the substrate.
3. In Sequoia, reduced male gametophyte is called pollen grain and its endosperm represents future sporophytes.
4. Wolffia is the smallest flowering plant.
Which of the following is not related to the floral formula of belladonna?
1.
2.
3.
4. K(5)
___(i)___ cambium forms complete, continuous but wavy ring during secondary growth in ___(ii)___
| 1. | (i) Vascular (ii) Dicot stem |
| 2. | (i) Cork (ii) Dicot leaf |
| 3. | (i) Vascular (ii) Dicot root |
| 4. | (i) Cork (ii) Dicot root |
Read the following statement with respect to prokaryotic cell
| I. | Single envelope system present. |
| II. | Mesosome increases surface area for enzyme action. |
| III. | The small circular DNA outside the genomic DNA confers unique phenotype. |
| IV. | Contains pseudovacuoles. |
1. Only I is correct.
2. Only I and II are correct.
3. Only I, II and III are correct.
4. I, II, III and IV are correct.
Crossing over
| 1. | Occurs in dyad stage of meiosis. |
| 2. | Causes recombination between genes of homologous chromosomes. |
| 3. | Occurs after chiasmata formation in leptotene. |
| 4. | Occurs in diplotene, the longest phase of meiosis. |
Loss of water in liquid phase from the margin of leaves in many herbaceous plants is/occurs
1. Guttation
2. Transpiration
3. At noon
4. Due to cohesion-tension model of water movement.
The essential element which maintains the ribosome structure is
1. Responsible for carbohydrate translocation.
2. Needed during mitotic spindle formation.
3. Constituent of ring structure of chlorophyll.
4. Needed in the synthesis of auxin.
Read the following statements
| Statement - I : | Water splitting complex is located on the inner side of the thylakoid membrane. |
| Statement - II : | During chemiosmosis breakdown of proton gradient releases energy. |
| 1. | Both I and II are incorrect. |
| 2. | I is correct but II is incorrect. |
| 3. | I is incorrect but II is correct. |
| 4. | Both I and II are correct. |
Complete the given reaction of the cycle present in the mitochondrial matrix:
1. A—Succinic acid; B—Pyruvate dehydrogenase.
2. A—Malic acid; B—Transacetylase.
3. A—OAA; B—Citrate synthase.
4. A— -KGA; B Thiokinase.
In prokaryotes, semi-conservative DNA replication was first proved by using
1. Radioactive 35S and 32P isotope.
2. Heavy 15N isotope.
3. Vicia faba.
4. 3H isotope in thymine.
(i) The VNTR belongs to a class of satellite DNA referred to as __A__.
(ii) Regulation of lac operon by repressor is referred to as __B__regulation.
(iii) The secondary structure of tRNA looks like __C__
A B C
1. Minisatellite Positive L-shaped
2. Micro-satellite Negative Inverted L-shaped
3. Minisatellite Negative Clover-leaf
4. Micro-satellite Positive Clover-leaf
Which of the following factors increases population density more significantly if new habitat is colonized?
1. Natality
2. Mortality
3. Immigration
4. Emigration
The natural reservoir for phosphorus is
1. Ocean
2. Rock
3. Atmosphere
4. Living organisms
A major disadvantage of external fertilization would be:
1. Lack of synchrony between sexes
2. Vulnerability of offspring to predators
3. Decreased amount of variations in the population
4. Limited number of offspring produced
Malignant tertian malaria is caused by Plasmodium:
1. vivax
2. malariae
3. falciparum
4. ovale
The type of joint between the carpal bones is:
1. Ball and Socket
2. Hinge
3. Pivot
4. Gliding
Atrial systole increases the flow of blood into the ventricles by about:
1. 10 %
2. 30 %
3. 50 %
4. 70 %
Consider the following method of breeding in animals:
I. Matings take place between same breed
II. No common ancestor is present on either side for 4 – 6 generations
III. It is the best method to improve milk production in cattle
This method of breeding is called:
1. Inter-specific hybridization
2. Cross-breeding
3. Inbreeding
4. Out-crossing
Select the correct statement from the ones given below with respect to dihybrid cross.
1. Tightly linked genes on the same chromosomes show very few recombinations.
2. Tightly linked genes on the same chromosome show higher recombinations.
3. Genes far apart on the same chromosome show very few recombinations.
4. Genes loosely linked on the same chromosome show similar recombinations as the tightly linked
ones.
Which one of the following cannot be explained on the basis of Mendel’s Law of Dominance?
1. Factors occur in pairs.
2. The discrete unit controlling a particular character is called a factor.
3. Out of one pair of factors one is dominant, and the other is recessive.
4. Alleles do not show any blending and both the characters recover as such in F2 generation.
What happens, when a cell is placed in a isotonic solution?
(a) There is not net flow of water towards the inside or outside.
(b) Cells are said to be flaccid.
(c) becomes positive.
(d) Enlargement and extension growth of cells occur.
The correct statement(s) is/are
1. a only
2. a and b only
3. a, b and c only
4. all are correct
Which of the following complex of mitochondrial ETS possess two copper centers?
1. Cytochrome b complex.
2. NADH dehydrogenase complex
3. Succinate dehydrogenase complex
4. Cytochrome c oxidase complex
Which of the following is not a hereditary disease -
1. Haemophilia
2. Cretinism
3. Cystic fibrosis
4. Thalasasemia
The given table enlists various hormones and their chemical nature.
Hormone Chemical composition
(1) ___(i)____ Peptide
(2) Testosterone ___(ii)____
(3) Thyroxine ___(iii)____
(4) ___(iv)____ Amino-acid derivative
Which is the correct option that completes the given table?
1. i-Cortisol; ii-Steroids; iii-Polypeptide; iv-Estradiol
2. i-Insulin; ii-Proteins; iii-Polypeptide, iv-Epinephrine
3. i-Cortical; ii-Proteins; iii-Iodothyronines; iv-Estradiol
4. i-Insulin; ii-Steroids; iii-Iodothyronines; iv-Epinephrine
In nematode resistance by RNA interference, some specific genes were introduced which form dsRNA. These were introduced in
1. E.coli
2. Host plant
3. Agrobacterium
4. Nematode
The C-peptide is:-
1. Not present in proinsulin
2. Present in mature insulin
3. Remove during maturation of insulin
4. Also present in artificial insulin
Catalytic converters are fitted into automobiles for reducing the emission of poisonous gases. Which of the following reaction does not take place in the catalytic converter?
1. Unburnt hydrocarbons - and water
2. Ozone -
3. Carbon monoxide -
4. Nitric oxide -
Which of the following is correct for the diagram given below?
1. (A) Dissolved oxygen, (B) Point of sewage discharge, (C) BOD
2. (A) BOD, (B) Point of treated water discharge, (C) Dissolved oxygen
3. (A) Dissolved oxygen, (B) point of treated water discharge, (C) BOD
4. (A) BOD, (B) Point of sewage discharge, (C) Dissolved oxygen
The thickness of ozone over poles changes with the season being lowest in
1. Antarctic spring
2. Polar autumn
3. Antarctic autumn
4. North hemisphere spring
In Duma's method of estimation of nitrogen, 0.35 g of an organic compound gave 55 ml of nitrogen collected at 300 K temperature and 715 mm pressure. The percentage composition of nitrogen in the compound would be:
(Aqueous tension at 300 K = 15 mm)
1. 16.45
2. 27.45
3. 44.45
4. 35.45
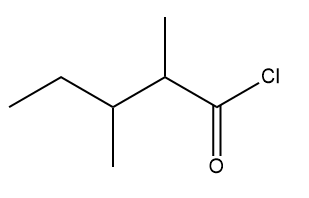
The IUPAC name of the above mentioned compound is -
1. 3, 4-Dimethylpentanoyl chloride
2. 1-Chloro-1-oxo-2,3-dimethylpentane
3. 2-Ethyl-3-methylbutanoylchloride
4. 2, 3-Dimethylpentanoyl chloride
The half-life time for the decomposition of a substance dissolved in is 2.5 hours at C. The amount of substance that will be left after 10 hours if the initial weight of the substance is 160 gm is:
| 1. | 20 gm | 2. | 30 gm |
| 3. | 40 gm | 4. | 10 gm |
Frenkel defect is not found in the halides of alkali metals because alkali metals have
1. high electropositivity
2. high ionic radii
3. high reactivity
4. ability to occupy interstitial sites
Litharge is not commonly used in:
1. manufacture of special glasses
2. glazing pottery
3. preparing paints
4. lead storage battery
The product formed on heating Pb(NO3)2
1.
2. Pb(NO2
3.
4.
For one mole of van der Waal's gas when b=0 and T=300 K, the PV vs. 1/V plot is shown below.
The value of the van der Waal's constant 'a' (atm. ) is-
1. 1.0
2. 4.5
3. 1.5
4. 3.0
At 298 K, assuming ideal behaviour, the average kinetic energy of a deuterium molecule is -
1. Two times that of a hydrogen molecule
2. Four times that of a hydrogen molecule
3. Half of that of a hydrogen molecule
4. Same as that of a hydrogen molecule
4.8 g of C(diamond) on complete combustion evolves 1584 kJ of heat. The standard heat of the formation of gaseous carbon is 725 kJ/mol. The energy required for the given process will be:
(i)
(ii)
| 1. | 725, 727 | 2. | 727, 725 |
| 3. | 725, 723 | 4. | None of the above |
One mole of a substance is cooled at the rate of as shown in the following graph. Curve AB, points B and C and curve CD represent respectively, the cooling of the liquid, start of freezing, completion of freezing and cooling of the solid. Based on this data, the entropy of fusion in is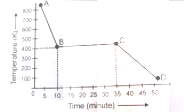
1. 10
2. 20
3. 25
4. 40
The isoelectric point of an amino acid is
1. The pH at which it exists in the acidic form
2. The pH at which exists in the basic form
3. The pH at which it exists in the Zwitterion form
4. The pH which is equal to its pKa value
A 500 g toothpaste sample has 0.4 g fluoride concentration. The fluoride concentration in terms of ppm will be -
| 1. | 200 ppm | 2. | 400 ppm |
| 3. | 500 ppm | 4. | 800 ppm |
In the transition of an electron in an atom, its kinetic energy changes from y to . The change in potential energy will be -
1. \(\frac{-3}{8} y\)
2. \({{3}\over{2}}y\)
3. \({{3}\over{4}}y\)
4. \(-{{3}\over{4}}y\)
The charge on the electron and proton are reduced to of their original values.
Let the present value of the Rydberg constant of H-atom be R, then the new value of the Rydberg constant will be
According to Crystal Field Theory (CFT), the compound, among the following, that forms a colored complex is:
1. [Ti(H2O)6]4+
2. [Cr(NH3)6]3+
3. K[VF6]
4. (NH4)2[TiCl6]
Among the following, the surfactant that will form micelles in aqueous solution at the highest molar concentration
1.
2.
3.
4.
Which of the following have non-zero dipole moment?
1.
2.
3.
4.
The group of metals having the positive value of electron gain enthalpy is:
| 1. | Cr, Fe | 2. | Mn, Zn |
| 3. | Fe, Zn | 4. | Cr, Mn |
The correctly matched option is:-
1. : Order of atomic radius
2. O > C > B > N : Order of ionisation energy
3. : Increasing order of Zeff
4. O < N < F < Ne : Order of electron affinity
What will be the freezing point of 0.2 molal aqueous solution of MgB ? lf salt show 60% dissociation in solution and Kf for water is 1.86K Kg mol-1
1.
2.
3.
4.
lf in daniel cell Kc = then cell the cell reaction will be :-
1. E=0.059 x 6; Spontaneous
2. E= 6 x 0.059; Nonspontaneous
3. E= -0.059 x 6; Nonspontaneous
4. E = -0.059 x 6; Spontaneous
Which of the following has highest value of pH:-
1. 0.1 M NaCl
2. 0.1 M NHCl
3. 0.1 M
4. 0.1 M
In alkaline medium oxidises to and it self gets reduces to . How many moles of are oxidised by 1 mol of ?
1. 1.5
2. 1
3. 2.5
4. 3.5
Sulphide ores are common for the metals :-
1. Ag, Cu and Pb
2. Ag, Cu and Sn
3. Ag, Mg and Pb
4. Al, Cu and Pb
On heating, ammonium dichromate and barium azide separately, we get :-
1. with ammonium dichromate and with barium azide
2. with ammonium dichromate and with barium azide
3. with ammonium dichromate and with barium azide
4. in both cases
Which of the following polymers has ethylene glycol as one of the monomer units?
1. 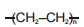
2. 
3. 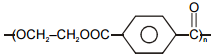
4. 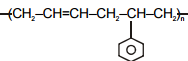
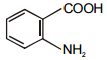
In the given reaction :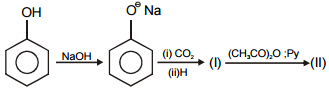
Identify (I) and (II) in the above reaction.
1. 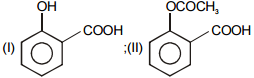
2. 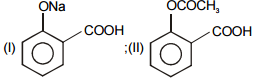
3. 
4. 
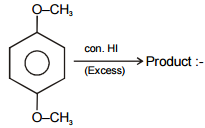
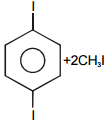
Consider the following reaction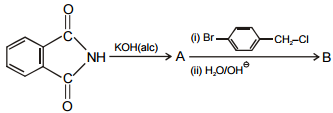
The product 'B' will be :-
1. 
2. 
3. 
4. 
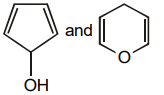
1. 
2. 
3. 
4. 
For the indicator the ratio is 7.0 at pH of 4.3 for the indicator will be :-
1. 3.5 10
2. 3.5
3. 3.5
4. 3.5
22.4 L of CO obtained at NTP by complete decomposition of 4 mol CaCO sample. Percentage purity of sample is :-
1. 50%
2. 25%
3. 75%
4. 100%
Which of the following salt directly give metal on heating :-
1.
2.
3. Pb(NO3)2
4. AgNO
BeCl + LiAlH
1. X is lithium hydride
2. X is BeH
3. X is BeCl . 2HO
4. X is LiH
Which of the following can act as an oxidizing agent?
1.
2.
3.
4. All
17.4 % (mass / volume) solution at 27°C is isotonic to 5.85% (mass / volume) NaCl solution at 27°C. If NaCl is 100% ionised, what is % ionisation of in aq. solution? [At wt. of K = 39, Na = 23 ]
1. 25%
2. 75%
3. 50%
4. None of these
The -labelled ester
is hydrolyzed with aqueous . The products will be
1.
2.
3.
4.
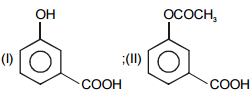
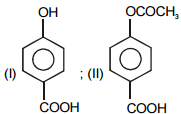
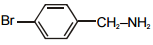
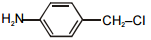
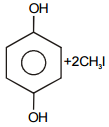
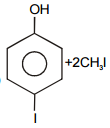
The pair of structures that does not represent isomers is:
| 1. | 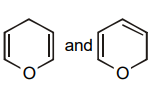 |
2. |  |
| 3. | 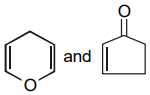 |
4. | 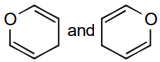 |
Magnesium allylide is _____, which on treatment with water gives _____
1. allylene
2. Propyne
3. Acetylene
4. Methane
Consider the following cell reaction
2Fe(s) + (g) + 4(aq) 2(aq) + 2(l)
E° = 1.67 V, At [] = 10 M, = 0.1 atm and pH = 3, the cell potential at 25 °C is :
1. 1.27 V
2. 1.77 V
3. 1.87 V
4. 1.57 V
ZSM-5 is used to convert:
1. Alcohol to petrol
2. Benzene to Toluene
3. Toluene to Benzene
4. Heptane to Toluene
The IUPAC name of the give compounds
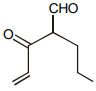
1. 3 – keto – 2 – methyl – pent – 4 – enal
2. 2 – keto – 2 – ethyl but – 2 – enal
3. 3 – keto – 2 – propyl pent – 4 – enal
4. 2 – keto – 2 – propyl pent – 4 – enal
The rate constant for two different reactions are e–2000/T and e–1000/T, respectively. The temperature at which is
1. 2000 K
2.
3. 1000 K
4.
In presence of a strong oxidizing agent is oxidized to arsinate and sulphate. What is the factor of
1. 4
2. 18
3. 28
4. 24
Which of the following hydrogen halides react with AgNO3 to give a precipitate that dissolves in hyposolution?
(I) HCl
(II) HF
(III) HI
(IV) HBr
1. (III), (I), (II)
2. (I), (III), (IV)
3. (IV), (II), (I)
4. (II), (IV), (III)
The extraction of silver is achieved by the initial complexation of the ore (Argentine) with X followed by reduction with Y. X and Y respectively are
1. and Zn
2. and Cu
3. and Zn
4. and Zn
A charged metallic conductor of arbitrary shape is positively charged. The conductor contains a cavity of arbitrary shape. Then
1. negative charges are induced on the surface of the cavity
2. electric field exists inside the cavity
3. the electric field in the cavity is zero
4. the electric field in the cavity depends on its position in the conductor
A student performs an experiment for determination of where l 1 m and he commits an error of . For T, he takes the time of n oscillations with the stop watch of least count . For which of the following data, the measurement of g will be most accurate:-
n
a. 5 mm 0.2 s 10
b. 5 mm 0.2 s 20
c. 5 mm 0.1 s 20
d. 5 mm 0.1 s 50
1. (a)
2. (b)
3. (c)
4. (d)
Two point charges placed at a certain distance r in the air, exert force F on each other. The distance r’ at which these charges will experience the same force in a medium of dielectric constant K is:
1.
2.
3.
4. rK
If the electric flux entering and leaving an enclosed surface respectively is , the electric charge inside the surface will be
1.
2.
3.
4.
Graph of velocity versus displacement of a particle moving in a straight line is shown in figure. The acceleration of the particle is:-
1. constant
2. increases linearly with x
3. increases parabolically with x
4. none of these
Find out the value of resistance R in figure:- 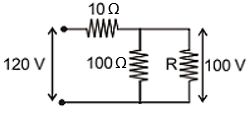
1.
2.
3.
4.

| 1. | \({V_1}\) increases. |
| 2. | \({V_2}\) increases. |
| 3. | \({V_2}\) & \({V_3}\) both increases. |
| 4. | one of \(V_2\) & \({V_3}\) increases and \({V_1}\) decreases. |
A man runs along a horizontal road holding his umbrella vertical in order to afford maximum protection from the rain. The rain is actually:
1. Falling vertical
2. Coming from front of the man
3. Coming from the back of the man
4. Either of (1), (2), or (3)
Three ships A, B and C are in motion. The motion of A as seen by B is with speed v towards north-east. The motion of B as seen by C is with speed v toward the north-west. Then as seen by A, C will be moving towards:-
1. north
2. south
3. east
4. west
A force is applied to an 8.0 kg object initially at rest. The magnitude of the net force varies with time as shown. What is the speed of the object at t=15 s.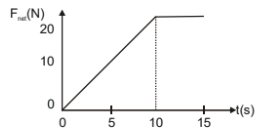
1. 10.0 m/s
2. 12.5 m/s
3. 25.0 m/s
4. 50.0 m/s
A capacitor of capacitance C, which is initially charged up to a potential difference , is connected with a battery of emf such that the positive terminal of the battery is connected with the positive plate of capacitor. Find the heat loss in the circuit during the process of charging.
1.
2.
3.
4. zero
A block of mass lies on a smooth horizontal table and is connected to another freely hanging block of mass by a light inextensible string passing over a smooth fixed pulley situated at the edge of the table. Initially the system is at rest with at a distance d from the pulley. Then the time taken for to reach the pulley is:-
1.
2.
3.
4. None of these
Figure shows a small loop carrying a current I. The curved portion is an arc of a circle of radius R and the straight portion is a chord to the same circle subtending an angle at the center. The magnetic induction at centre O is :-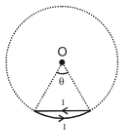
1. zero
2. always inward irrespective of the value of
3. inward as long as is less than
4. always outward irrespective of the value of
A magnetic needle suspended by a silk thread is vibrating in the Earth's magnetic field. If the temperature of the needle is increased, then:-
1. The time period decreases
2. The time period remains unchanged
3. The time period increases
4. The time periods first decreases then increases
A block is released from rest from a height h=5 m. After traveling through the smooth curved surface it moves on the rough horizontal surface through a length l=8 m and climbs onto the other smooth curved surface through a height h'. If , find h':-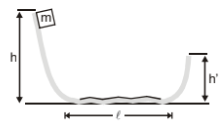
1. 2 m
2. 3 m
3. 1 m
4. zero
A boy weighing 50 kg finished long jump at a distance of 8 m. Considering that he moved along a parabolic path and his angle of jump was , his initial KE will be:-
1. 960 J
2. 1560 J
3. 2460 J
4. 1960 J
When a big hole is made in a magnet, then its magnetic moment becomes:
1. More
2. Less
3. Same
4. Zero
A pair of adjacent coils has a mutual inductance of 1.5 H. If the current in one coil changes from 0 to 20 A is 0.5 s, what is the change of flux linkage with the other coil?
1. 3 Wb
2. 3 mWb
3. 30 Wb
4. 30 mWb
In a magnetic field of 0.05 T, area of a coil changes from without changing the resistance which is . The amount of charge that flows during this period is:-
1.
2.
3.
4.
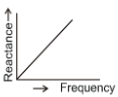
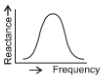
Which of the following plots may represent the reactance of a series L-C combination?
1. 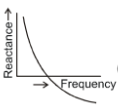
2. 
3. 
4. 
A mass of 10g moving horizontally with a velocity of 100 cm/s strikes a pendulum bob of mass 10g. Length of string is 50 cm. The two masses stick together. The maximum height reached by the system now is 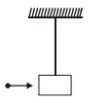
1. 7.5 cm
2. 5 cm
3. 2.5 cm
4. 1.25 cm
Four point masses each of value m are placed at the corners of a square ABCD of side l. The moment of inertia of this system about an axis passing through A and parallel to BD is:-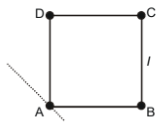
1.
2.
3.
4.
A small ball strikes a stationary uniform rod which is free to rotate in gravity-free space. The ball does not stick to the rod. The rod will rotate about-
1. its centre of mass.
2. the centre of mass of 'rod plus ball'
3. the point of impact of the ball on the rod.
4. the point about which the moment of inertia of the 'rod plus ball' is minimum.
Under some conditions, a material can be heated above or cooled below the normal phase change temperature without a phase change occurring. The resulting state:-
1. may be stable
2. may be unstable
3. must be stable
4. must be unstable
PV versus T graph of equal masses of is shown in figure. Choose the correct alternative:-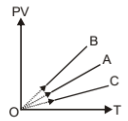
1. A corresponds to
2. A corresponds to He, B to and C to
3. C corresponds to He, B to
4. A corresponds to
A gas at pressure is contained in a vessel. If the masses of all the molecules are halved and their speeds doubled, the resulting pressure would be-
1.
2.
3.
4.
A ring consisting of two parts ADB and ACB of same heat conductivity k, is conducting an amount of heat H. The ADB part is now replaced with another metal keeping the temperature constant. The heat carried increases to 2H. what should be the heat conductivity of the new ADB part? Given 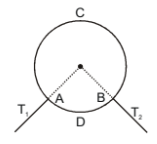
1.
2. 2k
3.
4. 3k
A Carnot engine is made to work first between and then between . The ratio of efficiency of the engine in two cases is:
1. 1:1.5
2. 1:1
3. 1:3
4. 1.73:1
A plane mirror is placed horizontally inside water . A ray falls normally on it. Then mirror is rotated through an angle . The minimum value of for which ray does not come out of the water surface is:-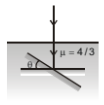
1.
2.
3.
4.
If there had been one eye of a man, then:
| 1. | image of the object would have been inverted |
| 2. | visible region would have decreased |
| 3. | image would have not been seen in three dimensional |
| 4. | Both (2) and (3) |
The two polaroids are crossed to each other. Now, one of them is rotated through . The percentage of incident unpolarized light that will pass through the system will be:-
1. 37.5%
2. 25%
3. 15%
4. 12.5%
With an increase in temperature, the viscosity of:
1. gases decreases and liquids increases.
2. gases increases and liquids decreases.
3. both gases and liquids increases.
4. both gases and liquids decrease.
For a given material, Young's modulus is 2.4 times the modulus of rigidity. Its Poisson's ratio is-
1. 0.1
2. 0.15
3. 0.2
4. 0.4
Small droplets of liquid are usually more spherical in shape than larger drops of the same liquid because:-
1. force of surface tension is equal and opposite to the force of gravity
2. force of surface tension predominates the force of gravity
3. force of gravity predominates the force of surface tension
4. force of gravity and force of surface tension act in the same direction and are equal
The collector plate in an experiment on photoelectric effect is kept vertically above the emitter plate. Light source is on and a saturation photo current is recorded. An electric field is switched on which has a vertically downward direction. Then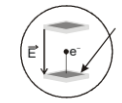
1. The photo current will increase
2. The kinetic energy of the electrons will increase
3. The stopping potential will decrease
4. The threshold wavelength will increase
The acceleration of a body due to the earth (radius R) at a distance 2R from the surface of the earth is (g=acceleration due to gravity at the surface of the earth):-
1.
2.
3.
4. g
If alpha, beta and gamma rays carry the same momentum, which has the longest wavelength?
| 1. | Alpha rays | 2. | Beta rays |
| 3. | Gamma rays | 4. | None, all have same wavelength |
All the surface are smooth and system is oscillating with amplitude A. Find extension of spring of spring constant when block is at extreme position:-
1.
2.
3. A
4.
The binding energies of the nuclei A and B are , respectively. Four atoms of the element B fuse to give one atom of element A and an energy Q is released. Then and Q related as:-
1.
2.
3.
4.
The maximum velocity of an electron emitted by light of wavelength incident on the surface of a metal of work function is
(a) (b)
(c) (d)
Where h = Planck's constant, m = mass of electron and c = speed of light.
A pure semiconductor has equal electron and hole concentration of \(10^{16}~\text{m}^{-3}\). Doping by indium increases \(n_h\) to \(4.5\times10^{22}~\text{m}^{-3}\).
What is \(n_e \) in the doped semiconductor?
1. \(10^{6}~\text{m}^{-3}\)
2. \(10^{22}~\text{m}^{-3}\)
3. \(\dfrac{10^{32}}{4.5\times10^{22}}~\text{m}^{-3}\)
4. \(4.5\times10^{22}~\text{m}^{-3}\)
A bus is moving with a velocity of 5 m/s towards a huge wall. The driver sounds a horn at a frequency of 165 Hz. If the speed of sound in air is equal to 335 m/s, the number of beats heard per second by a passenger on the bus will be:
| 1. | 3 | 2. | 4 |
| 3. | 5 | 4. | 6 |
The logic circuit given in diagram performs the logic operation:-
1. Y=AC
2.
3.
4.
In a p-n junction the depletion layer of thickness m has potential difference across it 0.2 V. The average electric field is (V/m):-
1.
2.
3.
4.
A particle is placed at rest inside a rough hollow hemisphere . The maximum height upto which the particle can remain stationary is:-
1.
2.
3.
4.