IUPAC name of given compound is:

1. 5-Methylhexanol
2. 2-Methylhexanol
3. 2-Methylhex-3-enol
4. 4-Methylpent-2-enol
The IUPAC name of the given compound is:
1. 4-Butylpenta-2,4-dienal
2. 5-Vinyloct-3-en-1-al
3. 5-Vinyloct-5-en-8-al
4. 3-Butyl-1,4-hexadien-6-al
But-2-ene exhibits cis-trans-isomerism due to
1. rotation around C2-C3 double bond
2. rotation around C3-C4 sigma bond
3. rotation around C1-C2 bond
4. restricted rotation around C=C bond
If optical rotation produced by the compound (i) is +52° then that produced by that the compound (ii) is:
1. -52°
2. +52°
3. 0°
4. unpredictable
Which of the following pairs are enantiomers?
1. 
2. 
3. 
4. 
In the following, the most stable conformation of n-butane is
1. 
2. 
3. 
4. 
Which of the following molecules exhibits chirality?
1. 2-Methylhexane
2. 3-Methylhexane
3. Neopentane
4. Isopentane
The property by virtue of which a compound can turn the plane of polarization of light is known as:
1. Photolysis
2. Phosphorescence
3. Optical activity
4. Polarization
What is the decreasing order of acidity for the compounds given below?

| 1. | III>IV>I>II | 2. | I>IV>III>II |
| 3. | II>I>III>IV | 4. | IV>III>I>II |
The correct increasing order of carbon-carbon bond length in the given compounds is:
1. III<II<I<IV
2. IV<I<II<III
3. I<II<III<IV
4. I<IV<III<II
The configuration of given tartaric acid is

1. 2R, 3R 2. 2R, 3S
3. 2S, 3S 4. 2S, 3R
Which of the following statements about the inductive effect is correct?
| 1. | The inductive effect transfers electrons from one carbon atom to another. |
| 2. | The inductive effect operates in both \(\sigma\)- and \(\pi\)-bonds. |
| 3. | The inductive effect does not create any charge in the molecule. |
| 4. | The inductive effect creates partial charges and is distance-dependent. |
The IUPAC name of the given compound is:
1. 2-Phenylpropan-3-al
2. Formylethylbenzene
3. 2-Phenylpropanal
4. Ethylformylbenzene
The IUPAC name of the given compound is:
1. Ethanoic propanoic anhydride
2. Propanoic ethanoic anhydride
3. 1-Ethanoyloxypropanone
4. 3-Ethanoyloxypropan-3-one
The IUPAC name of the given-below compound is:
1. Butane-2,3-dial
2. Butane-1,3-dione
3. Butane-2,3-dione
4. 1, 2-Dimethylethanedione
The IUPAC name of 
1. 4-Bromo benzenamine
2. 4-Amino-1-bromobenzene
3. 4-Bromo benzenamide
4. 1-Bromo benzencarboxamide
The IUPAC name of 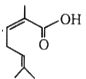 is:
is:
1. 2, 6-Dimethylhepta-2, 5-dienoic acid
2. 3, 7-Dimethylhepta-2, 5-dienoic acid
3. 1-Hydroxy-2, 6-dimethylhepta-2, 5-dienone
4. None of the above
The most stable carbocation among the following is:
| 1. |  |
2. |  |
| 3. | 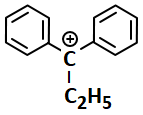 |
4. |  |
The electromeric effect in organic compounds is a
1. Temporary effect
2. Permanent effect
3. Electromeric effect is only observed in inorganic molecules
4. None of the above
The stability of 2,3-dimethyl but-2-ene is more than 2-butene. This can be explained in terms of:
1. resonance
2. hyperconjugation
3. electromeric effect
4. inductive effect
(CH3)4N+ is neither an electrophile, nor a nucleophile because it:
1. does not have electron pair for donation as well as cannot attract electron pair
2. neither has electron pair available for donation nor can accommodate electron since all shells of N are fully occupied.
3. can act as Lewis acid and base
4. none of the above
Which of the following is an electrophilic reagent?
1. RO-
2. BF3
3. NH3
4. ROH
The-I effect is shown by:
1. -COOH
2. -CH3
3. -CH3CH2
4. -CHR2
Sulphur trioxide act as:
1. An electrophile
2. A nucleophile
3. A homolytic agent
4. A base
Allyl isocyanide has:
1. 9 σ, and 4π-bonds.
2. 8 σ, and 5π-bonds
3. 9 σ, and 3π-bonds,
4. 8 σ, and 3 π bonds
Buta-1,3-diene and But-2-yne are:
1. Position isomers
2. Functional isomers
3. Chain isomers
4. Tautomers
Diastereomers can be separated by:
1. Fractional distillation
2. simple distillation
3. electrophoresis
4. all of these
In the following carbocations, the most stabile carbocation:
(1) RCH2C+H3
(2)
(3) 
(4)
The number of optical isomers of pent-3-en-2-ol is:
1. 2
2. 4
3. 8
4. 16
Geometrical isomerism is caused by-
1. Restricted rotation around C=C bond.
2. The presence of one asymmetric carbon atom.
3. The different groups attached to the same functional group.
4. None of the above.
Which of the following is optically active?
1. Alanine
2. 2-butanol
3. Lactic acid
4. All of these
Benzaldoxime exists in how many forms?
1. 1
2. 2
3. 3
4. 4
Nitroethane can exhibit one of the following kind of isomerism:
1. metamerism
2. optical activity
3. tautomerism
4. position isomerism
Number of possible isomers of glucose are:
1. 10
2. 14
3. 16
4. 20
Isomerism among compounds due to migration of a proton is known as:
1. Geometrical
2. Optical
3. Tautomerism
4. Position
The maximum number of stereoisomers possible for 3-hydroxy-2-methyl butanoic acid is:
1. 1 2. 2
3. 3 4. 4
Maleic and fumaric acids are:
1. tautomers
2. geometrical isomers
3. chain isomers
4. functional isomers
Which one among the following is most reactive towards nucleophilic addition reaction?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
which one of the following has the most acidic nature?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which of the following amines is the most basic
1. 
3. 4.
Which of the following is the most stable carbocation?
1. 

3. 4.
The non-aromatic compound among the following is:
1. 
3. 
Which of the following does not show tautomerism?
1. 2.
3. 
Major product of the following reaction is
Major product P is
1. 2.
3. 4.
Which compound will decarboxylate most easily upon heating in acidic medium?
1. 
3. 4.