What is meant by the "species problem"?
(1) The exact definition of species is still controversial.
(2) The exact mechanisms of speciation is not known.
(3) The number of species present is much more than identified species.
(4) The extinction of species is taking place at an alarming rate.
Which of the following taxonomic categories are correctly matched to their standard termination of names with respect to biological classification of plants?
| I | Division: | - phyta |
| II | Class: | - opsida |
| III | Order: | - ales |
| IV | Family: | - idae |
1. I, II, IV
2. I, II, III
3. II, III, IV
4. I, II, III, IV
The Indian Botanical Garden and the National Botanical Research Institute are located respectively at:
(1) Pune and Howrah
(2) Howrah and Lucknow
(3) Darjeeling and Lucknow
(4) Shimla and Dehradun
Match each item in Column I with one in Column II and choose your answer from the codes given below:
|
COLUMN I |
|
COLUMN II |
|
| I. | Flora | 1. | Useful in providing information for identification of names of species found in an area |
| II. | Manuals | 2. | Contains the actual account of habitat and distribution of plants of a given area. |
| III. | Monographs | 3. | contain information on any one taxon |
Codes:
|
I |
II |
III | |
| (1) |
1 |
2 |
3 |
| (2) | 3 | 2 | 1 |
| (3) | 2 | 1 | 3 |
| (4) | 2 | 3 | 1 |
Cell is the fundamental structural and functional unit of all living organisms because:
| 1. | Anything less than a complete structure of a cell does not ensure independent living. |
| 2. | The metabolic reactions can only occur inside a living cell. |
| 3. | Nucleic acids present in the cells ensure living state. |
| 4. | The membrane bound organelles cause differentiation for proper functioning. |
Prokaryotic mesosomes do not help in:
1. Cell wall formation
2. DNA replication
3. Respiration
4. Photosynthesis
The endomembrane system of a eukaryotic cell does not include:
1. Endoplasmic reticulum
2. Lysosome
3. Vacuole
4. Peroxisome
The cis and trans faces of the Golgi Apparatus are:
| 1. | Similar but not interconnected |
| 2. | Similar and interconnected |
| 3. | Entirely different but interconnected |
| 4. | Entirely different and not interconnected |
Plastids used in storing proteins are called as:
1. Amyloplasts
2. Aleuroplasts
3. Elaioplasts
4. Chromoplasts
A centriole-like structure, from which the cilium and the flagellum emerge, is the:
1. Centrosome
2. Basal body
3. Axoneme
4. Vimentin
The outer membrane of the nucleus:
I. is continuous with the Endoplasmic reticulum
II. does not bear ribosomes.
1. Only I is correct
2. Only II is correct
3. Both I and II are correct
4. Both I and II are incorrect
The centrioles replicate during:
1. G1 phase
2. S phase
3. G2 phase
4. Early prophase
Bivalent chromosomes clearly appear as tetrads during:
1. Zygotene
2. Pachytene
3. Diplotene
4. Diakinesis
The beginning of diplotene is recognized by:
1. appearance of recombination nodules
2. crossing over
3. dissolution of synaptonemal complex
4. appearance of chiasmata
The portion of human stomach into which the oesophagus opens is the:-
1. Cardiac
2. Fundus
3. Corpus
4. Pylorus
Muscularis layer of the wall of the alimentary canal is formed by:-
1. Smooth muscle arranged into an inner longitudinal and an outer circular layer
2. Smooth muscle arranged into an inner circular and an outer longitudinal layer
3. Skeletal muscle arranged into an inner longitudinal and an outer circular layer
4. Skeletal muscle arranged into an inner circular and an outer longitudinal layer
The structural and functional units of the human liver are:-
1. Glisson capsules
2. Hepatic lobes
3. Hepatic Iobules
4. Hepatic sinusoids
The optimum pH for the enzyme salivary amylase is:
1. 4.5
2. 6.8
3. 7.3
4. 8.2
Intrinsic factor is secreted by the_cells of the human stomach.
1. Mucus neck cells
2. Chief cells
3. Oxyntic cells
4. G-cells
Lipases can be activated by:
1. Enterokinase
2. Enterogastrone
3. Chyme
4. Bile
Which of the following is a sub-mucosal gland?
1. Oxyntic glands of stomach
2. Crypts of Lieberkuhn of small intestine
3. Brunner's glands of the duodenum
4. Goblet cells
Meiosis I:
| 1. | is always followed by interphase |
| 2. | is not followed by any period of rest |
| 3. | is followed by a period of interkinesis |
| 4. | is sometimes followed by interphase |
In guard cell when sugar is converted into starch, the stomatal pore
1. opens partially
2. opens completely
3. closes completely
4. Remains unchanged
The lipid component of the plasma membrane mainly consists of:
1. Triglycerides
2. Cholesterol
3. Phosphoglycerides
4 Proteins
The endomembrane system of a eukaryotic cell includes all except:
1. Vacuoles
2. Lysosomes
3. Peroxisomes
4. Golgi complex
Carotenoid pigments are found in:
| I: | Chloroplasts |
| II: | Chromoplasts |
| III: | Leucoplasts |
1. Only I
2. Only I and II
3. Only II
4. I, II and III
Microbodies, which help to convert stored lipids into carbohydrates so they can be used for plant growth, are known as:
1. Peroxisomes
2. Sphaerosomes
3. Glyoxysomes
4. Lysosomes
If the DNA content of an onion tip cell is 2C at the end of the M-phase, what would be its DNA content at the end of the S-phase?
1. C
2. 2C
3. 3C
4. 4C
There cannot be a living state without :
1. Metabolism
2. Capability of reproduction
3. Self-consciousness
4. Presence of DNA in a cell
All the following are the part of a eukaryotic cell endomembranous system except:
1. Vacuole
2. Lysosome
3. Golgi complex
4. Mitochondria
The cell shown in the given diagram is in:
1. Early prophase
2. Late prophase
3. Metaphase
4. Telophase
Unless specifically stained by a vital stain like Janus Green, which of the following will not be visible under a microscope?
1. Rough endoplasmic reticulum
2. Smooth endoplasmic reticulum
3. Golgi apparatus
4. Mitochondria
Fat soluble pigments like xanthophylls are present in:
1. Chromoplasts
2. Aleuroplasts
3. Elaioplasts
4. Amyloplasts
Proteins that are to be used outside the cell are synthesized:
| 1. | in the mitochondria |
| 2. | on the rough endoplasmic reticulum |
| 3. | on the smooth endoplasmic reticulum |
| 4. | on free ribosomes |
The two types of cellular organelles that transform energy are:
| 1. | Chromoplasts and Leucoplasts |
| 2. | Mitochondria and Chloroplasts |
| 3. | Mitochondria and Endoplasmic reticulum |
| 4. | Chloroplasts and Golgi apparatus |
The arrangement of outer and central microtubules in a cilium is called the:
1. 9 + 1 pattern
2. 9 + 0 pattern
3. Flagellin pattern
4. 9 + 2 pattern
When a cell stops growing, say due to shortage of nutrients, this will occur in which phase of the cell cycle?
1.
2.
3. S
4.
At what phase of meiosis are homologous chromosomes separated ?
1. Prophase l
2. Prophase ll
3. Anaphase l
4. Anaphase ll
Match each item in Column I with one item in Column II and choose your answer from the codes given below :
|
Column I |
Column II |
||
|
I. |
Zygotene |
1. |
Crossing over |
|
II. |
Pachytene |
2. |
Chaismata formation |
|
III. |
Diplotene |
3. |
Bivalent formation |
|
IV. |
Diakinesis |
4. |
Terminalization of chaismata |
Codes :
|
I |
II |
III |
IV |
|
|
1. |
1 |
2 |
3 |
4 |
|
2. |
2 |
1 |
4 |
3 |
|
3. |
3 |
1 |
2 |
4 |
|
4. |
3 |
2 |
1 |
3 |
Consider the following cellular functions :
| A: | Transport of substances |
| B: | Synthesis of proteins |
| C: | Synthesis of steroidal hormones |
| D: | Glycogen metabolism |
| E: | Detoxification of drugs |
| F: | Giving rise to sphaerosomes |
1. two
2. four
3. six
4. five
The eukaryotic cell structure, which controls the activities of organelles and also plays a major role in heredity is :
1. Nucleoli
2. Nucleus
3. Nucleoid
4. Mitochondria
Select the incorrect statement :
| 1. | Leeuwenhoek first described a live cell |
| 2. | Robert Brown discovered the nucleus |
| 3. | Robert Hooke first saw a living cell |
| 4. | Schwann proposed that bodies of animals and plants are composed of cells and products of cells |
" One of the most important functions of the plasma membrane is the transport of molecules."Which of the following is correct with respect to this statement ?
| 1. | Polar molecules can pass easily through the lipid bilayer |
| 2. | The membrane does not support passive transport |
| 3. | Neutral solutes require a carrier protein of membrane for transport |
| 4. | The membrane is selectively permeable to some molecules present on either side of it |
Read the following statements
| A: | Mitochondria help in generation of adenosine triphosphate |
| B: | The outer mitochondrial membrane folds into several cristae |
| 1. | Only B is incorrect |
| 2. | Both A and B are correct |
| 3. | Only A is incorrect |
| 4. | Both A and B are incorrect |
The function like delivery of packaged material either to the intracellular targets or secreted outside the cell is principally performed by :
1. Lysosomes
2. Golgi body
3. Vacuole
4. ER
Mitochondria and plastids are similiar in all of the following aspects, except :
| 1. | Possess circular ds-DNA |
| 2. | Have endo-symbiotic origin |
| 3. | Are the sites of carbohydrate bio-synthesis |
| 4. | Divide by fission |
Select the incorrect match :
| 1. | Thylakoids - contain chlorophyll pigments |
| 2. | Chromoplasts - contain xanthophylls |
| 3. | Leucoplast- Stores tannin and resin |
| 4. | Food vacuoles - Have food particles |
Lysosomes are :
| 1. | Double membrane bound vesicular structures |
| 2. | Rich in few hydrolases |
| 3. | Formed by packaging in Golgi apparatus |
| 4. | Formed by engulfing the food particles |
Choose the correct statement with respect to the following diagram:
| 1. | Section of flagella showing different parts seen in eukaryotes and prokaryotes. |
| 2. | It shows the cartwheel organisation with 9 + 0 microtubular arrangement. |
| 3. | They form structures which cause movement. |
| 4. | It forms structure which is not covered by any membrane. |
Chromosome with one shorter arm and no longer arm, having centromere slightly away from middle is :
1. Acrocentric
2. Metacentric
3. Sub-metacentric
4. Telocentric
How many of the following features are associated, with the endomembrane system ?
Synthesis of steroidal hormones; Digestion of food, storage of waste, storage of proteins; synthesis of ATP; formation of glycolipid
1. Four
2. Three
3. Five
4. Two
Select the odd one out with respect to ribosomes :
| 1. | Synthesised inside the nucleus in eukaryotes |
| 2. | Composed of DNA and proteins |
| 3. | Not surrounded by any membrane |
| 4. | Prokaryotic ribosomes are 70 S |
Select the correct statement :
| 1. | Centrioles form the basal body of cilia and flagella |
| 2. | Secondary constrictions on the chromosomes do not have a constant location |
| 3. | Microbodies are present in plant cells only |
| 4. | Nucleoli are less in number in cells undergoing protein synthesis |
The membrane bound space found in the cytoplasm :
| 1. | Is active site for ribosomal RNA synthesis |
| 2. | Helps in protein synthesis |
| 3. | Contains water, sap, excretory product |
| 4. | Forms the spindle apparatus |
Select the incorrect match :
| 1. | Cristae - Increase the surface area |
| 2. | Amyloplast - stores starch |
| 3. | Inner nuclear membrane - Bears ribosomes |
| 4. | Kinetochore - Attachment site for spindle fibre |
Which of the following is not membrane bound cell organelle present in the cell of mango plant ?
1. Mitochondria
2. Chloroplast
3. Ribosome
4. Centriole
Find the incorrect statement:
| 1. | Middle lamella is mainly made up of calcium pectate |
| 2. | Cell wall is formed on the inner side of the cell therefore secondary wall formed first |
| 3. | Middle lamella glues the neighbouring cells together |
| 4. | Cell wall helps in cell to cell interaction and provides barrier to undesirable macromolecules |
Transitional vesicles enclosing biochemicals pinched off from ER fuse with :
| 1. | Maturing face of golgi body |
| 2. | Forming face of golgi body |
| 3. | Primary lysosome for intracellular digestion |
| 4. | Plasma membrane to remove them from cell |
According to fluid mosaic model of plasma membrane the quasifluid nature of :
| 1. | Protein enables lateral movement of lipids within the overall bilayer |
| 2. | Lipid and protein enable the lateral movement of carbohydrates |
| 3. | Lipid enables lateral movement of proteins within the overall bilayer |
| 4. | Lipid enables the flip-flop movement of proteins |
Cell theory formulated by Schleiden and Schwann does not explain one of the following features ?
1. Organisms are composed of cells and their products
2. All cells are basically alike in their structure and metabolism
3. New cells originate from pre-existing cells
4. The functions of an organism is an outcome of activities and interactions of its constituent cells
The enzymes present in lysosomes belong the class ____ and acidic condition inside the lysosome is maintained by ___:
| 1. | Oxidoreductases, pumping protons |
| 2. | Hydrolases, secretion of acids |
| 3. | Hydrolases, pumping of protons |
| 4. | Lyases, removal of hydroxyl ions |
Which of the following feature is common in all types of plastids?
1. Double membrane
2. Presence of chlorophyll
3. Storage nature
4. Presence of linear DNA
A nuclear pore allows:
| 1. | Unidirectional movement of DNA |
| 2. | RNA movement only |
| 3. | RNA and protein movement |
| 4. | Protein movement only |
Correct statement in relation to vacuoles is :
| 1. | It is a triple membrane bound space found in cytoplasm containing sap |
| 2. | It can occupy 90% of cell volume in plants |
| 3. | Its membrane allows transport of materials along the concentration gradient only |
| 4. | Concentration of ions is significantly lesser in vacuole than cytoplasm |
Glycosylation, general secretion and recycling of broken plasma membrane are functions specifically performed by:
1. Glyoxysome
2. SER
3. RER
4. Golgi complex
Which of the following membrane proteins are partially or totally buried in cell membrane?
1. Integral proteins
2. Peripheral proteins
3. Both
4. Glycoproteins
Fluid mosaic model was given by Singer and Nicolson in the year _________
1. 1972
2. 1976
3. 1982
4. 1986
The ability of proteins to move laterally within the membrane is measured as
1. mobility
2. flexibility
3. fluidity
4. none
Functions of cell wall in eukaryotic cells
| 1. | give shape to cell |
| 2. | prevent from mechanical damage |
| 3. | protect from infection |
| 4. | all |
Which of the following walls is capable of growth in a plant cell?
| 1. | Primary wall | 2. | Secondary wall |
| 3. | Both (1) & (2) | 4. | Middle lamella |
In 30 S and 40 S ribosomes, 'S' stands for
1. sub-unit
2. Svedberg's unit
3. single unit
4. size
Select correct matching :
|
Column l |
Column ll |
|
(Type of leucoplast) |
(Stored food) |
|
A. Amyloplast |
1. Oil and fat |
|
B. Elaioplast |
2. Protein |
|
C. Aleuroplasts |
3. Carbohydrate |
1. A-3, B-1, C-2
2. A-1, B-2, C-3
3. A-3, B-2, C-1
4. A-2, B-3, C-1
Outer membrane of nucleus remains continuous with which cell organelle
1. ER
2. Golgi body
3. Lysosome
4. All
Perinuclear space is about
1. 1-5 nm
2. 5-10 nm
3. 10-50 nm
4. > 100 nm
RER is frequently seen in cells associated with frequent synthesis and secretion of
1. lipid
2. glucose
3. protein
4. all
Amount or number of mitochondria in a cell depends on
| 1. | anatomical structure of cell |
| 2. | size of cell |
| 3. | colour and contour of the cell |
| 4. | physiological activity of cell |
How is new mitochondria formed by pre-existing ones?
1. Mitosis
2. Fission
3. Conjugation
4. Budding
A elaborate network of filamentous proteinaceous structures present in the cytoplasm is collectively known as
1. Cilia
2. Flagella
3. Cytoskeleton
4. ER
The central part of the centriole is proteinaceous and is known as
1. Spokes
2. Doublet
3. Hub
4. Linkers
Which of the following is not modified mucosal epithelium?
1. Goblet cells
2. Brunner’s glands
3. Crypts of Lieberkuhn
4. Gastric glands
Identify the stages of cell cycle in which the following events occur
(A) Duplication of most of the organelles
(B) Duplication of centrioles
(C) Synthesis of tubulin proteins
(A) (B) (C)
1. G2 phase G1 phase M phase
2. S phase M phase G0 phase
3. M phase Interphase G1 phase
4. G1 phase S phase G2 phase
Trypsinogen can be activated by:
1. HCl
2. Enterogastrone
3. Trypsin
4. Bicarbonate ions
Consider the characters of a certain digestive enzyme:
I. It is a proteolytic enzyme secreted as zymogen.
II. It acts in an alkaline pH.
III. It activates many pancreatic enzymes.
The digestive enzyme is:
1. Pepsin
2. Enterokinase
3. Cholecystokinin
4. Trypsin
The number of species that are known and described ranges between
(1) 1.2 – 1.3 million
(2) 1.2 – 1.3 billion
(3) 1.7 – 1.8 million
(4) 1.7 – 1.8 billion
Cell division occurs _____ in plants and ______ in animals
(a) Continuously, only upto a certain age
(b) Only upto a certain age, continuously
(c) Continuously, never
(d) Once, twice
Match the entities in Column I with Column II
Column I Column II
A. Planaria I. Binary fission
B. Fungi II. Asexual pores
C. Yeast III. Budding
D. Hydra IV. True regeneration
E. Amoeba V. Fragmentation
F. Species Plantarum VI. Linnaeus
and Systema Naturae
(a) A- I, B - II, C -III, D - IV, E -VI, F -V
(b) A - IV, B - II, V, C - III, D - III, E - I, F - VI
(c) A - III, B - II, C - IV, D - III, E - VI, F - I
(d) A - II, B - III, C - I, D - IV, E - II, F - VI
Which of the following organisms do not reproduce?
(a) Mules
(b) Sterile worker bees
(c) Both (a) and (b)
(d) None
Metabolic reactions take place
1. In isolated cell-free systems
2. In living systems
3. Both (1) and (2)
4. Either (1) or (2)
Identify the following diagram :-
| 1. | Transition to Metaphase (mitosis). |
| 2. | Transition to Anaphase II (meiosis). |
| 3. | Transition to Metaphase I (meiosis). |
| 4. | Transition to Anaphase (mitosis). |
How many meiotic division are required to form 300 seeds of wheat?
1. 250
2. 350
3. 75
4. 375
Mn(aq) + 8H+(aq) + 5e- Mn2+(aq) + 4H2O(l);=1051V
Cr2O72-(aq) +14H+(aq) + 6e 2Cr3+(aq) + 7H2O(l);=1.38V
Fe3+(aq) + e- Fe2+(aq);=0.77V
Cl2(g) + 2e- 2Cl-(aq); = 1.40V
The incorrect statement regarding the quantitative estimation of aqueous Fe(NO3)2 is -
1. Mn can be used in aqueous HCl
2. Cr2O72- can be used in aqueous HCl
3. MnO-24 can be used in aqueous H2SO4
4. Cr2O72- can be used in aqueous H2SO4
When aqueous NaCl solution is electrolysed using inert electrodes then pH of the solution-
1. Increases.
2. Decreases.
3. Remains same.
4. First increases then decreases.
What is the current efficiency of an electrode deposition of Cu metal from CuSO4 solution in which 9.8 gm copper is deposited by the passage of 5 amperes current for 2 hours?
1. 41.4 %
2. 50%
3. 75%
4. 82.8 %
In the electrolysis of which solution, OH ions are discharged in preference to Cl ions?
1. Dilute NaCl
2. Very dilute NaCl
3. Fused NaCl
4. Solid NaCl
During electrolysis, 2A current is passed through an electrolytic solution for 965 s. The number of moles of electrons passed will be
1. 0.02
2. 0.01
3. 200
4. 0.037
. For this cell is negative
1.
2.
3.
4. None of these
The emf of the cell is 0.46 V.
Given = 0.80 v
1. -0.34 V
2. 1.26 V
3. -1.26 V
4. 0.34 V
20 g of chlorine are evolved in 6 hour from sodium chloride solution by the current of
1. 5 ampere
2. 10 ampere
3. 2.5 ampere
4. 50 ampere
What is the current efficiency of an electrode deposition of Cu metal from solution in which 9.8 gm copper is deposited by the passage of 5 ampere current for 2 hours ?
1. 41.4 %
2. 50%
3. 75 %
4. 82.8%
Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL. The molarity of the solution is:
1. 7.78 M
2. 2.00 M
3. 2.05 M
4. 2.22 M
Mole fraction of the solute in a 1.00 molal aqueous solution is
1. 1.7700
2. 0.1770
3. 0.0177
4. 0.0344
Which has highest freezing point?
1. 0.01 M NaCl
2. 0.05 M Urea
3. 0.01 M MgCl2
4. 0.02 M NaCl
for is 1.86 k/m. Therefore, the freezing point of 0.1 M NaCl solution in water is expected to be
When mercuric iodide is added to the aqueous solution of potassium iodide is
1. Freezing point is raised
2. Feezing point is lowered
3. Freezing point does not change
4. Boiling point does not change
A 5% solution of cane sugar (molar mass = 342) is isotonic with 1% of a solution of an unknown solute. The molar mass of unknown solute is
1. 136.2
2. 171.2
3. 68.4
4. 34.2
What is the freezing point of a solution contains 8.1 gm HBr in 100 gm water assuming the acid to be 90% ionized? (Kf for H2O = 1.86 k/m)
Which of the following solution has the highest equivalent conductance?
1. 0.01 M NaCl
2. 0.05 M NaCl
3. 0.005 M NaCl
4. 0.02 M NaCl
The resistance of 0.01 N solution of an electrolyte AB at 328K is 100 ohm. The specific conductance of solution is (cell constant = 1 cm-1)
1. 100 ohm
2. 10-2 ohm
3. 10-2 ohm-1cm-1
4. 102 ohm-cm
The increase in the molar conductivity of HCl with dilution is due to
1. Increase in the self ionisation of water
2. Hydrolysis of HCl
3. Decrease in the self ionisation of water
4. Decrease in the interionic forces
A solution of in water is electrolysed using pt electrodes. The products at the cathode and anode are respectively
Given that
The most reactive metal which displaces other metals from their salts in solution is
1. Ag
2. Cu
3. Co
4. Zn
The emf of the cell is 0.83V. The emf of this cell could be increased by
1. increasing the concentration of Tl+ ions
2. increasing the concentration of Cu2+ ions
3. increasing the concentration of both
4. None of the above
For a reaction has been found to be . The is
1. 0.354 V
2. 0.708 V
3. 0.0098 V
4. 1.36 V
The solution of nickel sulphate in which nickel rod dipped is diluted to 10 times. The potential of nickel
1. Decreases by 60 mV
2. Increases by 30 mV
3. Decreases by 30 mV
4. Decreases by 60 mV
Two electrodes are fitted in conductance cell 1.5 cm apart while the area of cross section of each electrode is 0.75 cm2. The cell constant is
1. 1.0125 cm
2. 0.5 cm
3. 2.0 cm-1
4. 0.2 cm-1
Pick out the incorrect statement
1. Equivalent conductance increases with dilution
2. Molar conductance increases with dilution
3. Specific conductance increases with dilution
4. Specific resistance increases with dilution
Aluminium phosphate is 100% ionized in 0.01m aqueous solution. Hence, is
1. 0.01 2. 0.015
3. 0.0175 4. 0.02
60 gm of urea is dissolved in 1100 gm solution. To keep as 1 mol/kg, amount of water separated in the form of ice will be
1. 40 gm 2. 60 gm
3. 100 gm 4. 200 gm
Which solution has the highest osmotic pressure?
1. 0.1 M NaCl 2. 0.5 M Urea
3. 0.1 M MgCl2 4. 0.2 M NaCl
An ideal solution is made by mixing 2 moles of benzene (Po = 266 mm) and 3 moles of another liquid (Po = 236 mm). The total vapour pressure of the solution at the same temperature would be
1. 502 mm 2. 248 mm
3. 600 mm 4. 258 mm
A binary solution of ethanol and n-heptane is an example of
1. Ideal solution
2. Non-ideal solution with +ve deviation
3. Non-ideal solution with -ve deviation
4. Unpredictable behaviour
0.5 M solution of urea is isotonic with
1. 0.5 M NaCl solution
2. 0.5 M sugar solution
3. 0.5 M BaCl2 solution
4. 0.5 M solution benzoic acid in benzene
What is the value of if for of a at
1.
2.
3.
4.
Which has maximum freezing point?
1. 200 ml of 2 M BaCl2 solution
2. 200 ml of 2 M CuSO4 solution
3. 200 ml of 2 M Glucose solution
4. All have same freezing point
A solution of a non-volatile solute in water has a boiling point of 375.3 K. The vapour pressure of water above this solution at 338 K is :- [Given (water)=0.2467 atm at 338 K and for water=0.52 K kg mol-1]
1. 0.18 atm
2. 0.23 atm
3. 0.34 atm
4. 0.42 atm
Which of the following changes decrease the vapour pressure of water kept in a sealed vessel?
1. Decreasing the quantity of water.
2. Increasing the quantity of water.
3. Decreasing the volume of the vessel to one-half.
4. Decreasing the temperature of water.
for the reaction is
1. 11.44 kJ
2. -180.8 kJ
3. -150.5 kJ
4. 28.5 kJ
At 20C, the vapour pressure of 0.1 M solution of urea is 0.0311 mm less than that of water and the vapour pressure of 0.1 M solution of KCl is 0.0574 mm less than that of water. The apparent degree of dissociation of KCl at this dilution is :-
1. 92.1%
2. 84.6%
3. 68.4%
4. 54.1%
What is the current efficiency of an electrode deposition of Cu metal from CuSO4 solution in which 9.8 gm copper is deposited by the passage of 5 amperes current for 2 hours?
1. 41.4%
2. 50%
3. 75%
4. 82.8%
Two liquids A and B have and in the ratio of 1 : 3 and the ratio of number of moles of A and B in liquid phase are 1 : 3 then mole fraction of 'A' in vapour phase in equilibrium with the solution is equal to:
1. 0.1
2. 0.2
3. 0.5
4. 1.0
The vapour pressure of acetone at is 185 torr. When 1.2 g of a non-volatile substance was dissolved in 100 g of acetone at , its vapour pressure was 183 torr. The molar mass (g/mol) of the substance is
1. 32
2. 64
3. 128
4. 488
Two faraday of electricity is passed through a solution of . The mass of copper deposited at the cathode is (at. mass of Cu=63.5 amu)
1. 0
2. 63.5 g
3. 2 g
4. 127 g
Higher order (>3) reaction are rare due to
1. low probability of simultaneous collision of all the reacting species
2. increase the entropy and activation energy is more molecules are involved
3. shifting of equilibrium towards the reactants due to elastic collision
4. loss of active species on collision
Assertion: An increase in surface area increases the rate of evaporation.
Reason: Stronger the inter-molecular attraction forces, faster is the rate of evaporation at a given temperature.
1.
2.
3.
4.
Monochromatic radiation emitted when an electron in a hydrogen atom jumps from the first excited state to the ground state irradiates a photosensitive material. The stopping potential is found to be 4V. The threshold wavelength of the material is
In a photoelectric experiment using a metal of work function 1.8 eV, if the maximum kinetic energy of emitted electrons is 1.5eV, then the corresponding value to stopping potential is:
1. 1.8V
2. 3.3V
3. 0.3V
4. 1.5V
When the light of wavelength is used in a photoelectric experiment, the maximum kinetic energy of emitted electrons is found to be K. What will the maximum kinetic energy of the photoelectrons when the light of wavelength is used instead of :
1. Zero
2. Non zero but less than 2 K
3. 2K
4. More than 2K
1. 2
2. 4
3.
4.
| 1. | The diffraction pattern is not observed on the screen in the case of electrons. |
| 2. | The angular width of the central maximum of the diffraction pattern will increase. |
| 3. | The angular width of the central maximum will decrease. |
| 4. | The angular width of the central maximum will remain the same. |
In Young's double-slit experiment, the slits are 0.4 mm apart and illuminated by photons of two wavelengths = 600 nm and = 700nm. At what minimum distance from the common center, bright fringe from one interference pattern coincides with bright fringe from other? (Given that the distance of the screen from the plane of slits is 80cm.)
1. 7.2 mm
2. 6.0 mm
3. 8.4 mm
4. 9.8 mm
The concept of matter-wave was given by:
1. de-Broglie
2. Einstein
3. Maxwell
4. Bohr
A ray of light will suffer total internal reflection if it-
1. goes from rare medium to denser medium
2. Incident at less than the critical angle
3. Strikes the interface normally
4. incident at an angle greater than critical angles
The shortest wavelength of Balmer series is about
1. 912
2. 121
3. 3648
4. 4864
When the light of wavelength is made to fall on metal in a photoelectric experiment, the maximum kinetic energy of emitted electrons is found to be 2.5eV. The work function of the metal is:
1. 2.5eV
2. 3.1eV
3. 0.9eV
4. 1.5eV
1.
2.
3.
4.
The angular speed of electron in a hydrogen atom in orbit is proportional to -
1.
2.
3.
4.
A diffraction pattern is observed using a beam of red light. What will happen if the red light is replaced by the blue light?
| 1. | No change takes place. |
| 2. | Diffraction bands become narrower. |
| 3. | Diffraction bands become broader. |
| 4. | Diffraction pattern disappears. |
The phenomenon of polarisation justify which nature of light?
1. longitudinal
2. transverse
3. both
4. geometrical
If the momentum of a photon is p, then its energy is :
1. pc
2.
3.
4.
For non-relativistic speeds, the wavelength associated with an electron and its kinetic energy E are related as:
1.
2.
3.
4.
An electron at rest is acceleration by a potential difference of 600V. The de-Broglie wavelength associated with the electron is:
1.
2.
3.
4.
The de-Broglie wavelength of a body of mass 1 kg moving with a velocity of 2000 m/s is
1.
2.
3.
4. None of these
Light of wavelength 4000 Å falls on a photosensitive metal and a negative 2V potential stops the emitted electrons.
The work function of the material (in eV) is approximately
1. 1.1
2. 2.0
3. 2.2
4. 3.1
If the wavelength of light used is halved and the numerical aperture of the compound microscope is doubled, then its resolving power will
1. Remain unchanged
2. Doubled
3. Halved
4. Quadrupled
In the situation shown, the incident monochromatic ray retraces its path after its incidence on the silvered surface. The speed of light inside the prism will be

1.
2.
3.
4.
When neutron moving with Kinetic Energy 2eV collides with
stationary in the ground state, the collision will be:
1. Must be elastic
2. may be inelastic
3. Both
4. Must be perfectly inelastic
The potential difference applied to an X-ray tube is 5 kV and current through it is 3.2 mA. The number of electrons striking the target per sec is:
1.
2.
3.
4.
The work functions for metal A, metal B, and metal C are respectively. Then-
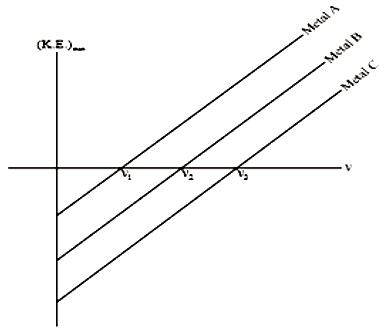

1.
2.
3.
4. None of these
| 1. | \(\frac{m_1}{m_2} \) | 2. | \(\frac{m_2}{m_1} \) |
| 3. | \(1 \) | 4. | \(\sqrt{\frac{\mathrm{m}_2}{\mathrm{~m}_1}}\) |
nth bright fringe of red light coincides with (n+1)th bright fringe of green light . The value of n =?
1. 4
2. 5
3. 3
4. 2
The ratio of the frequency of the long wavelength limits of the Lyman and Balmer series of hydrogen spectrum is:
1. \(27:5\)
2. \(5:27\)
3. \(4:1\)
4. \(1:4\)
The Rydberg constant R for hydrogen is
1.
2.
3.
4.
The electron emitted in beta radiation originates from
1. inner orbits of atoms
2. free electrons existing in nuclei
3. decay of neutron in a nucleus
4. photon escaping from the nucleus
The photoelectric work function for a metal surface is 4.125 eV. The cut off wavelength for this surface is
1. 4125
2. 2062.5
3. 3000
4. 6000
Carbon, Silicon, and Germanium atoms have four valence electrons each. Their valence and conduction bands are separated by energy gaps represented by \(\left(E_g\right)_C,(E_g)_{Si}~\text{and}~(E_g)_{Ge}\) respectively. Which one of the following relationships is true in their case?
1. \(\left(E_g\right)_C<\left(E_g\right)_{G e} \)
2. \(\left(E_g\right)_C>\left(E_g\right)_{S i} \)
3. \(\left(E_g\right)_C=\left(E_g\right)_{S i} \)
4. \(\left(E_g\right)_C<\left(E_g\right)_{S i}\)
A double slit of separation 1.5 mm is illuminated by white light (between 4500 -7000 ). On a screen 120 cm away coloured interference pattern is formed. If a pinhole is made on this screen at a distance 3.0 mm from the central white fringe, which of the following wavelength will be absent in the transmitted light?
1. 5000
2. 6000
3. 6500
4. 7000
A proton and an electron both have a kinetic energy of 1 eV. Which has the longer wavelength?
1. Electron
2. Proton
3. Both have the same wavelength
4. Wavelength and K.E. are not related
The de Broglie wavelength of an electron in the nth Bohr orbit is related to the radius R of the orbit as
1.
2.
3.
4.
Light of frequency 1.5 times the threshold frequency is incident on photo-sensitive material. If the frequency is halved and intensity is doubled, the photocurrent becomes
1. Quadrupled
2. Doubled
3. Halved
4. Zero
A nuclear reaction along with the masses of, the particle taking part in it is as follows
A + B C + D + QMeV
1.002 1.004 1.001 1.003 Q
amu amu amu amu amu
The energy Q liberated in the reaction is
1. 1.234 MeV
2. 0.931 MeV
3. 0.465 MeV
4. 1.862 MeV
The ratio of the radii of the nuclei and is approximately
1. 6 : 10
2. 13 : 52
3. 40 : 177
4. 14 : 73
The mass defect for the nucleus of helium is 0.0303 a.m.u. What is the binding energy per nucleon for helium in MeV?
1. 28
2. 7
3. 4
4. 1
The mass density of a nucleus varies with mass number A as
1.
2.
3. Constant
4.
A stationary radioactive nucleus of mass 210 units disintegrates into an alpha particle of mass 4 units and residual nucleus of mass 206 units. If the kinetic energy of the alpha particle is E, the kinetic energy of the residual nucleus is
1.
2.
3.
4.
After 2 hours of the initial amount of a certain radioactive isotope remains undecayed. The half-life of isotope is
1. 60 minutes
2. 45 minutes
3. 30 minutes
4. 15 minutes
The radioactivity of a given sample of whisky due to tritium (half life 12.5 years) was found to be only 3% of that measured in a recently purchased bottle marked “7 year old”. The sample must have been prepared about
1. 220 years back
2. 300 years back
3. 400 years back
4. 70 years back
In the reaction , if the binding energies of and are respectively a, b and c (in MeV), then the energy (in MeV) released in this reaction is
1.
2.
3.
4.
Fission of nuclei is possible because the binding energy per nucleon in them
1. Increases with mass number at low mass numbers
2. Decreases with mass number at low mass numbers
3. Increases with mass number at high mass number
4. Decreases with mass number at high mass number